1477-42-5
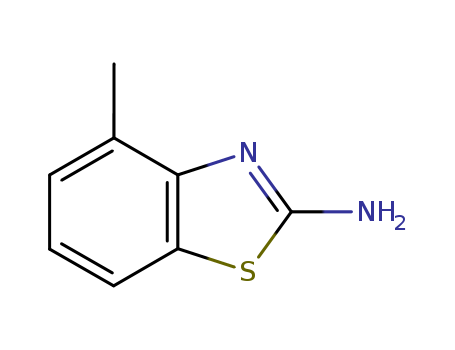
- Product Name:2-Amino-4-methylbenzothiazole
- Molecular Formula:C8H8N2S
- Purity:99%
- Molecular Weight:164.231
Product Details;
CasNo: 1477-42-5
Molecular Formula: C8H8N2S
Appearance: White powder
Purity 99% Min 2-Amino-4-methylbenzothiazole 1477-42-5 Spot Supply with Safe Transportation
- Molecular Formula:C8H8N2S
- Molecular Weight:164.231
- Appearance/Colour:White powder
- Vapor Pressure:0.000287mmHg at 25°C
- Melting Point:137-139 °C(lit.)
- Refractive Index:1.726
- Boiling Point:322 °C at 760 mmHg
- PKA:pK1: 4.7(+1) (25°C)
- Flash Point:148.6 °C
- PSA:67.15000
- Density:1.315 g/cm3
- LogP:2.76810
2-Amino-4-methylbenzothiazole(Cas 1477-42-5) Usage
|
Air & Water Reactions |
2-Amino-4-methylbenzothiazole is sensitive to moisture. Insoluble in water. |
|
Reactivity Profile |
An amine, organosulfide. Organosulfides are incompatible with acids, diazo and azo compounds, halocarbons, isocyanates, aldehydes, alkali metals, nitrides, hydrides, and other strong reducing agents. Reactions with these materials generate heat and in many cases hydrogen gas. Many of these compounds may liberate hydrogen sulfide upon decomposition or reaction with an acid. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. |
|
Health Hazard |
ACUTE/CHRONIC HAZARDS: When heated to decomposition 2-Amino-4-methylbenzothiazole emits very toxic fumes of SOx and NOx. |
|
Fire Hazard |
Flash point data for 2-Amino-4-methylbenzothiazole are not available. 2-Amino-4-methylbenzothiazole is probably combustible. |
|
General Description |
White powder. |
InChI:InChI=1/C8H8N2S/c1-5-3-2-4-6-7(5)10-8(9)11-6/h2-4H,1H3,(H2,9,10)
1477-42-5 Relevant articles
Studies on phenothiazines. VII. (1). Synthesis of 3-substituted 2-aminobenzenethiols and their conversion into phenothiazines
Gupta,Ojha,Kumar
, p. 1325 - 1327 (1980)
-
ETUDE DE LA FRAGMENTATION PAR IMPACT ELECTRONIQUE DE DERIVES DU BENZOTHIAZOLE
Claude, Saturnin,Tabacchi, Raffaele,Duc, Laurent,Fuchs, Rudolf,Boosen, Karl-Josef
, p. 682 - 692 (1980)
The mass spectra of eighteen substituted...
Method for synthesizing 4-methyl-2-benzothiazolehydrazine
-
Paragraph 0048; 0057; 0059; 0061; 0063; 0065; 0067; 0069, (2020/07/12)
The invention discloses a method for syn...
SN-Donor methylthioanilines and copper(II) complexes: Synthesis, spectral properties, and in vitro antimicrobial activity
Olalekan, Temitope E.,Ogunlaja, Adeniyi S.,Watkins, Gareth M.
, (2019/04/25)
Methylthioanilines, a series of sulfur-n...
Scalable synthesis and antibacterial evaluation of 2-(3-(N-(substituted phenyl)sulfamoyl)ureido)benzothiazoles
Cheraiet, Zinelaabidine,Meliani, Saida,Nessaib, Mounir,Hessainia, Sihem,Boukhari, Abbas,Djahoudi, Abdelghani,Regainia, Zine
, (2019/08/12)
A new series of 2-(3-(N-(substituted phe...
Preparation method of tricyclazole intermediate
-
Paragraph 0019-0023, (2018/03/26)
The invention relates to a preparation m...
1477-42-5 Process route
-
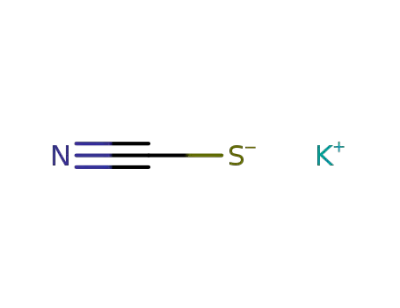
-
333-20-0
potassium thioacyanate

-
-
95-53-4
o-toluidine

-
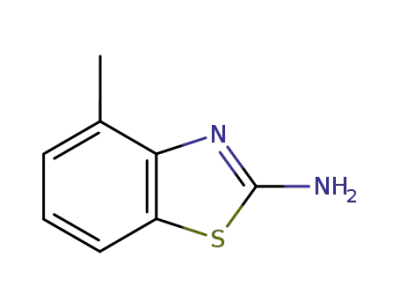
-
1477-42-5
4-methyl-1,3-benzothiazol-2-amine
| Conditions | Yield |
|---|---|
|
With
sodium iodide dichloride;
at 70 ℃;
for 2h;
|
91% |
|
potassium thioacyanate; o-toluidine;
With
nano-BF3/SiO2;
In
acetonitrile;
for 0.5h;
Cooling with ice;
With
bromine;
In
acetonitrile;
at 0 - 20 ℃;
for 5h;
|
89% |
|
With
bromine; acetic acid;
at 0 - 20 ℃;
for 13.75h;
|
|
|
With
bromine; acetic acid;
for 10h;
|
-
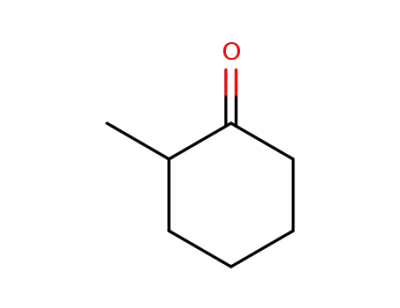
-
583-60-8
2-Methylcyclohexanone

-
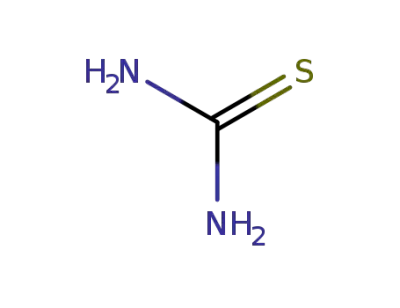
-
17356-08-0
thiourea

-

-
1477-42-5
4-methyl-1,3-benzothiazol-2-amine
| Conditions | Yield |
|---|---|
|
With
iodine; oxygen; toluene-4-sulfonamide;
In
dimethyl sulfoxide;
at 70 ℃;
for 24h;
under 760.051 Torr;
Reagent/catalyst;
Solvent;
Temperature;
Green chemistry;
|
84% |
|
With
iodine; oxygen; toluene-4-sulfonic acid;
In
dimethyl sulfoxide;
at 75 ℃;
for 24h;
under 760.051 Torr;
|
71% |
1477-42-5 Upstream products
-
2268-77-1
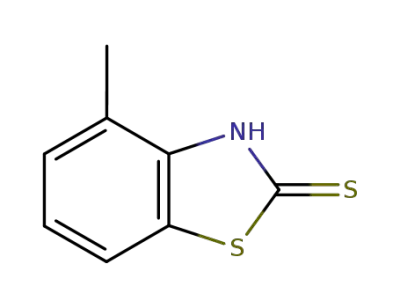
4-methylbenzo[d]thiazole-2(3H)-thione
-
614-78-8
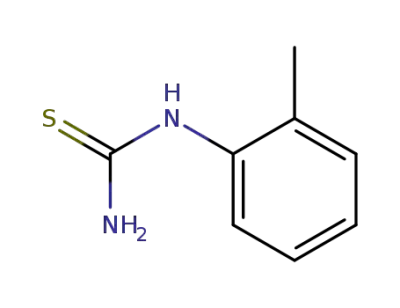
o-tolylthiourea
-
7791-25-5
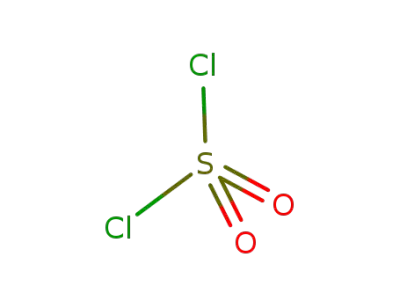
sulfuryl dichloride
-
108-90-7
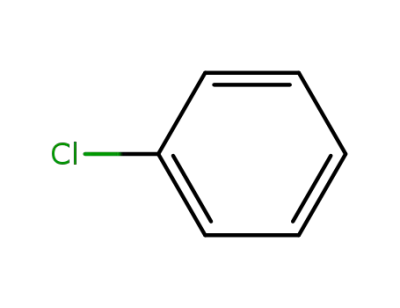
chlorobenzene
1477-42-5 Downstream products
-
1819-99-4
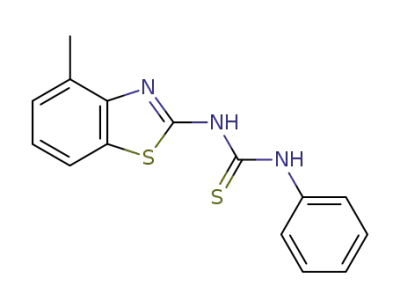
1-(4-methyl-benzothiazol-2-yl)-3-phenyl-thiourea
-
3622-32-0
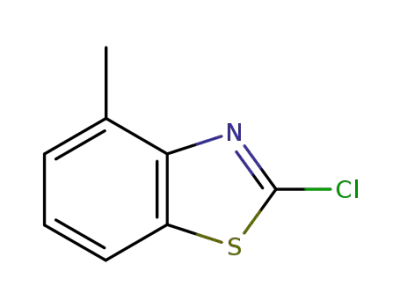
2-chloro-4-methyl-benzothiazole
-
1457-46-1
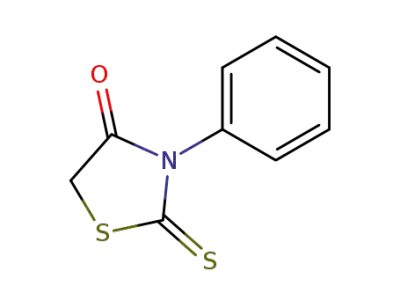
3-phenylrhodanine
-
117844-38-9
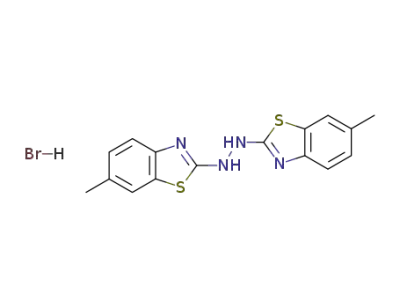
N,N'-Bis-(6-methyl-benzothiazol-2-yl)-hydrazine; hydrobromide
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
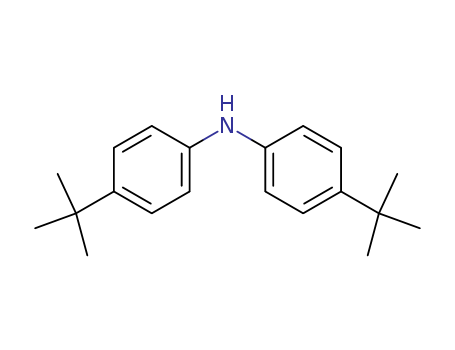
4,4'-DI-TERT-BUTYLDIPHENYLAMINE
CAS:4627-22-9
-
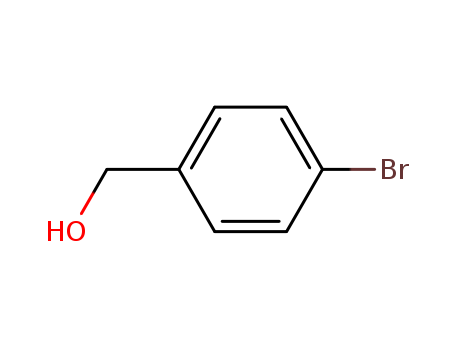
4-Bromobenzyl alcohol
CAS:873-75-6