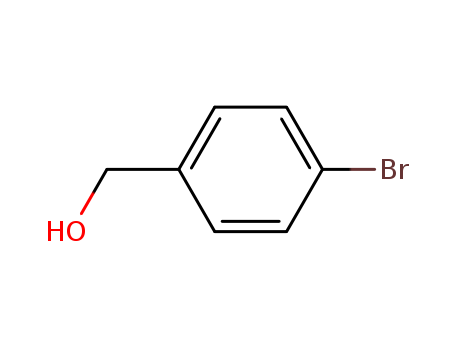
873-75-6
- Product Name:4-Bromobenzyl alcohol
- Molecular Formula:C7H7BrO
- Purity:99%
- Molecular Weight:187.036
Product Details;
CasNo: 873-75-6
Molecular Formula: C7H7BrO
Appearance: White to slightly beige crystal
Excellent chemical plant bulk supply 4-Bromobenzyl alcohol 873-75-6
- Molecular Formula:C7H7BrO
- Molecular Weight:187.036
- Appearance/Colour:White to slightly beige crystal
- Vapor Pressure:0.00397mmHg at 25°C
- Melting Point:75-77 °C
- Refractive Index:1.591
- Boiling Point:267.8 °C at 760 mmHg
- PKA:14.16±0.10(Predicted)
- Flash Point:115.7 °C
- PSA:20.23000
- Density:1.565 g/cm3
- LogP:1.94140
4-Bromobenzyl alcohol(Cas 873-75-6) Usage
|
Synthesis Reference(s) |
The Journal of Organic Chemistry, 48, p. 3074, 1983 DOI: 10.1021/jo00166a029Tetrahedron Letters, 36, p. 3169, 1995 DOI: 10.1016/0040-4039(95)00504-6 |
|
Flammability and Explosibility |
Notclassified |
|
General Description |
4-Bromobenzyl alcohol undergoes three-component reaction with acetylferrocene and arylboronic acid to give ferrocenyl ketones containing biaryls. It undergoes oxidation reaction in the presence of polyvinylpolypyrrolidone-supported hydrogen peroxide, silica sulfuric acid and ammonium bromide to yield 4-bromobenzaldehyde. It undergoes efficient trimethylsilylation reaction with 1,1,1,3,3,3-hexamethyldisilazane in the presence of catalytic amount of aspartic acid in acetonitrile. |
InChI:InChI=1/C7H7BrO/c8-7-3-1-6(5-9)2-4-7/h1-4,9H,5H2
873-75-6 Relevant articles
Green synthesis of metal oxide nanoparticles and their catalytic activity for the reduction of aldehydes
Muthuvinothini,Stella
, p. 48 - 56 (2019)
In the present work, a green synthesis o...
-
Gerrard,W.,Green,D.B.
, p. 91 - 96 (1967)
-
Hydrogenation of Esters by Manganese Catalysts
Li, Fu,Li, Xiao-Gen,Xiao, Li-Jun,Xie, Jian-Hua,Xu, Yue,Zhou, Qi-Lin
, (2022/01/13)
The hydrogenation of esters catalyzed by...
Unifying Scheme for the Biosynthesis of Acyl-Branched Sugars: Extended Substrate Scope of Thiamine-Dependent Enzymes
Clapés, Pere,Hernández, Karel,Krug, Leonhard,Müller, Michael,R?hr, Caroline,Steitz, Jan-Patrick,Walter, Lydia
supporting information, (2022/02/05)
Thiamine diphosphate (ThDP) dependent en...
CeO2-nanocubes as efficient and selective catalysts for the hydroboration of carbonyl groups
Bhawar, Ramesh,Bose, Shubhankar Kumar,Patil, Kiran S.
supporting information, p. 15028 - 15034 (2021/09/04)
The CeO2-nanoparticle catalysed hydrobor...
Experimental and density functional theory studies on hydroxymethylation of phenylboronic acids with paraformaldehyde over a Rh-PPh3 catalyst
Wang, Kuan,Lan, Jie,He, Zhen-Hong,Cao, Zhe,Wang, Weitao,Yang, Yang,Liu, Zhao-Tie
, (2020/12/01)
The synthesis of benzyl alcohols (BAs) i...
873-75-6 Process route
-
-
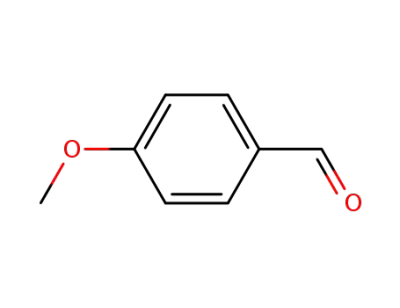
123-11-5
4-methoxy-benzaldehyde

-
-
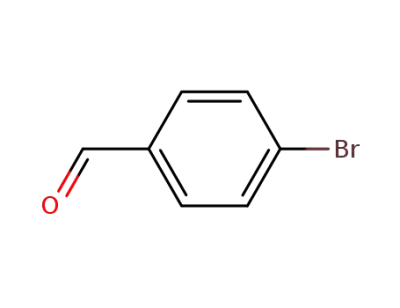
1122-91-4
4-bromo-benzaldehyde

-
-
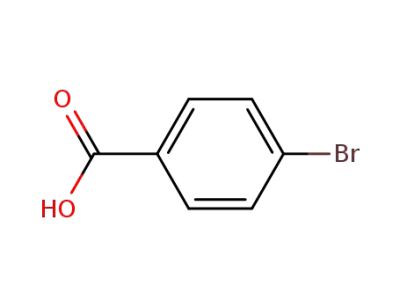
586-76-5
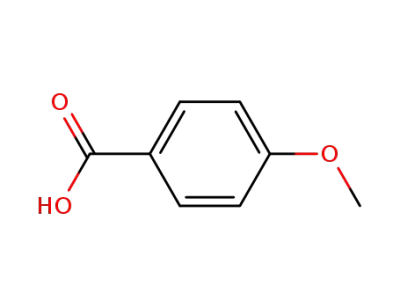
4-Bromobenzoic acid

-
-
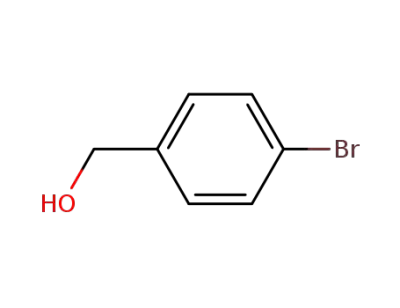
873-75-6
4-bromobenzenemethanol

-
-
100-09-4
4-methoxybenzoic acid

-
-
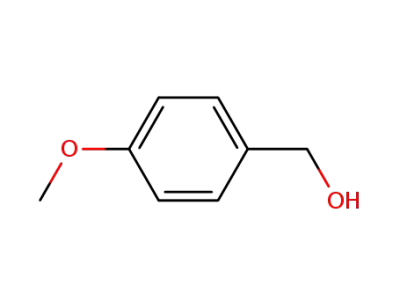
105-13-5
4-Methoxybenzyl alcohol
| Conditions | Yield |
|---|---|
|
|
-
-
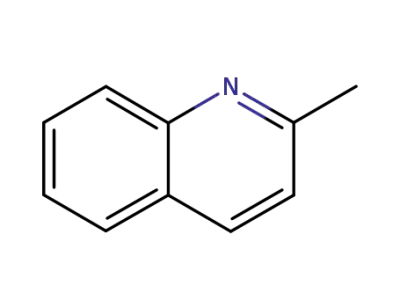
91-63-4
2-methylquinoline

-

-
1122-91-4
4-bromo-benzaldehyde

-

-
873-75-6
4-bromobenzenemethanol

-
-
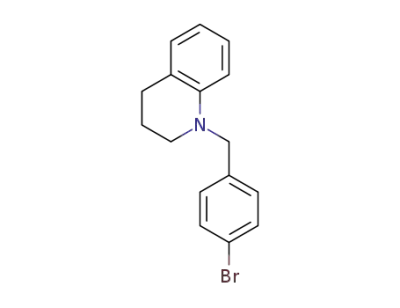
885438-74-4
N-(4-bromobenzyl)-1,2,3,4-tetrahydroquinaldine

-
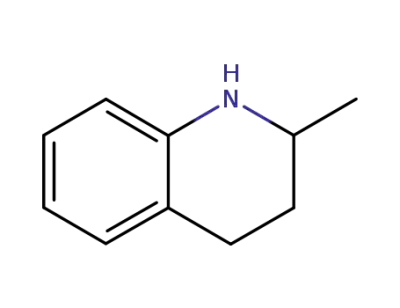
-
1780-19-4
1,2,3,4-tetrahydro-2-methylquinoline
| Conditions | Yield |
|---|---|
|
With
(7,8-benzoquinolinato)hydrido(aqua)bis(triphenylphosphine)iridium(III) tetrakis[3,5-bis(trifluoromethyl)phenyl]borate; hydrogen;
In
toluene;
at 80 ℃;
for 18h;
|
62% 33% 35% |
873-75-6 Upstream products
-
106-37-6
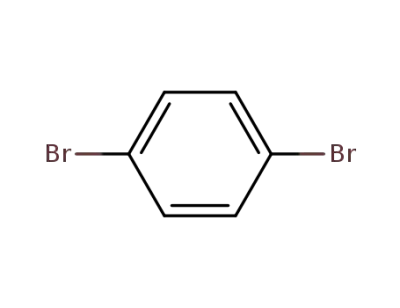
1.4-dibromobenzene
-
586-76-5

4-Bromobenzoic acid
-
21388-92-1
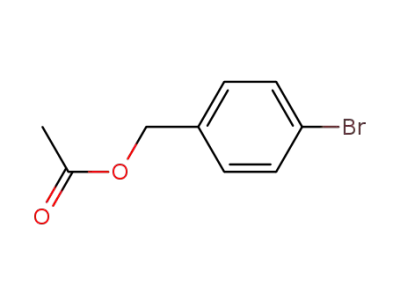
4-bromobenzyl acetate
-
1122-91-4

4-bromo-benzaldehyde
873-75-6 Downstream products
-
856181-41-4
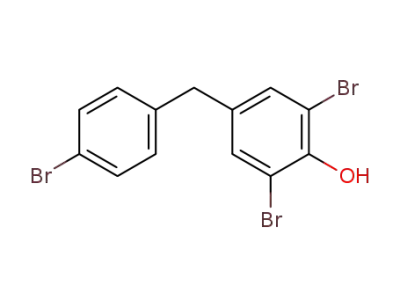
2,6-dibromo-4-(4-bromo-benzyl)-phenol
-
20614-06-6
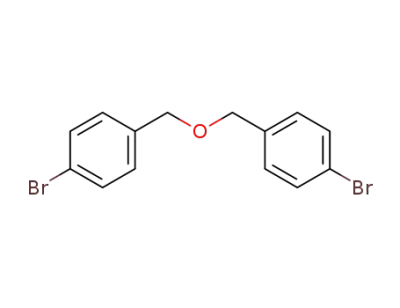
bis(4-bromobenzyl) ether
-
1122-91-4

4-bromo-benzaldehyde
-
589-17-3
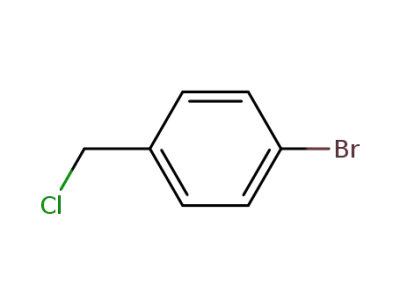
p-bromobenzyl chloride
Relevant Products
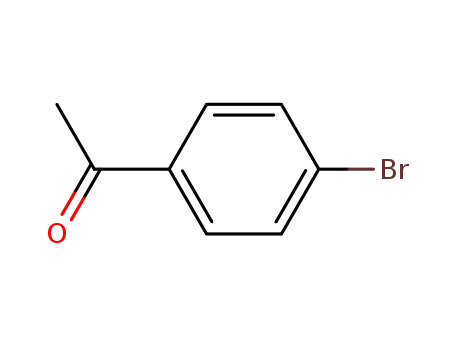
-
4'-Bromoacetophenone
CAS:99-90-1
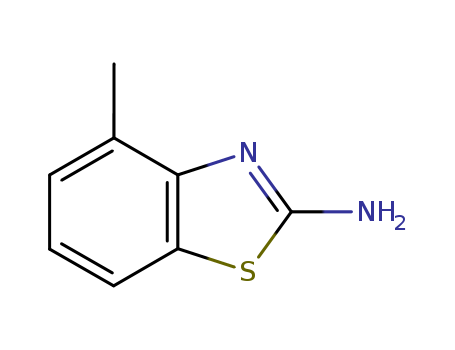
-
2-Amino-4-methylbenzothiazole
CAS:1477-42-5
-
DMT-2’-F-dA(Bz)-CE-Phosphoramidite
CAS:136834-22-5