4627-22-9
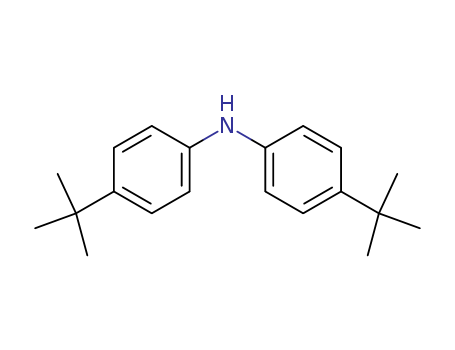
- Product Name:4,4'-DI-TERT-BUTYLDIPHENYLAMINE
- Molecular Formula:C20H27N
- Purity:99%
- Molecular Weight:281.441
Product Details;
CasNo: 4627-22-9
Molecular Formula: C20H27N
Factory Supply Industrial Grade 4,4'-DI-TERT-BUTYLDIPHENYLAMINE 4627-22-9 with Best Price
- Molecular Formula:C20H27N
- Molecular Weight:281.441
- Vapor Pressure:4.61E-06mmHg at 25°C
- Melting Point:108 °C
- Refractive Index:1.552
- Boiling Point:382.8 °C at 760 mmHg
- PKA:1.47±0.40(Predicted)
- Flash Point:188.1 °C
- PSA:12.03000
- Density:0.974 g/cm3
- LogP:6.09820
4,4'-DI-TERT-BUTYLDIPHENYLAMINE(Cas 4627-22-9) Usage
InChI:InChI=1/C20H27N/c1-19(2,3)15-7-11-17(12-8-15)21-18-13-9-16(10-14-18)20(4,5)6/h7-14,21H,1-6H3
4627-22-9 Relevant articles
Stable diarylnitroxide diradical with triplet ground state
Rajca, Andrzej,Shiraishi, Kouichi,Rajca, Suchada
, p. 4372 - 4374 (2009)
Nitroxide diradical 2, the first isolate...
Temperature-Dependent Effects of Alkyl Substitution on Diarylamine Antioxidant Reactivity
Shah, Ron,Poon, Jia-Fei,Haidasz, Evan A.,Pratt, Derek A.
, p. 6538 - 6550 (2021)
Alkylated diphenylamines are among the m...
Copper-Assisted Amination of Boronic Acids for Synthesis of Bulky Diarylamines: Experimental and DFT Study
Levitskiy, Oleg A.,Grishin, Yuri K.,Sentyurin, Vyacheslav V.,Magdesieva, Tatiana V.
, p. 12575 - 12584 (2017)
Comparative investigation of copper-assi...
Nitrogen-containing compound, organic electroluminescent device, and electronic device
-
Paragraph 0111-0115; 0118, (2021/01/24)
The invention provides a nitrogen-contai...
COMPOUND FOR ORGANIC ELECTRONIC ELEMENT, ORGANIC ELECTRONIC ELEMENT USING THE SAME, AND AN ELECTRONIC DEVICE THEREOF
-
Paragraph 0166-0174, (2021/03/09)
The present invention provides a novel c...
COMPOUND FOR ORGANIC ELECTRONIC ELEMENT, ORGANIC ELECTRONIC ELEMENT USING THE SAME, AND A ELECTRONIC DEVICE THEREOF
-
Paragraph 0103; 0106-0109, (2021/06/22)
In the present invention, provided is a ...
4627-22-9 Process route
-
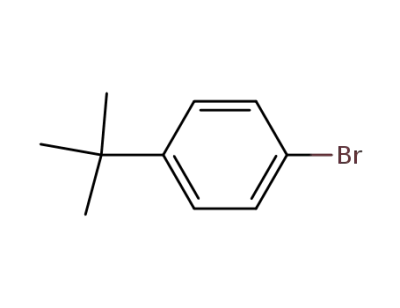
-
3972-65-4
1-bromo-4-tert-butylbenzene

-
-
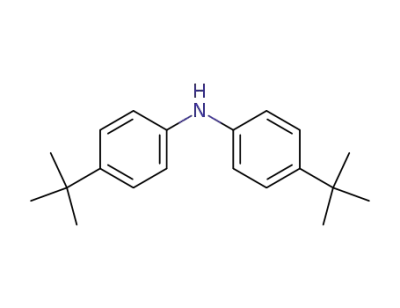
4627-22-9
bis(4-tert-butylphenyl)amine

-
-
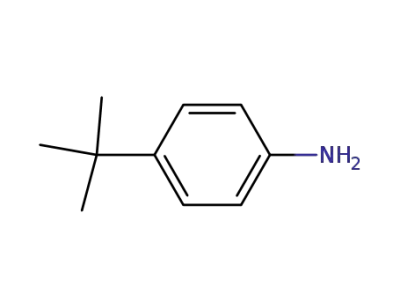
769-92-6
4-tert-Butylaniline
| Conditions | Yield |
|---|---|
|
With
ammonia; sodium t-butanolate;
(CyPF-t-Bu)PdCl2;
In
1,2-dimethoxyethane;
at 90 ℃;
for 24h;
under 4137.18 - 10343 Torr;
|
86% |
|
1-bromo-4-tert-butylbenzene;
With
ammonia; sodium t-butanolate;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 90 ℃;
for 24h;
under 4137.29 - 10343.2 Torr;
With
hydrogenchloride;
In
1,2-dimethoxyethane; water;
at 20 ℃;
for 0.0833333h;
With
sodium hydrogencarbonate;
In
1,2-dimethoxyethane; water;
Product distribution / selectivity;
|
86% |
|
With
lithium amide;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 80 ℃;
for 10h;
Product distribution / selectivity;
|
72% |
|
1-bromo-4-tert-butylbenzene;
With
lithium amide;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 80 ℃;
for 24h;
Sealed vial;
With
hydrogenchloride;
In
1,2-dimethoxyethane; water;
at 20 ℃;
for 0.0833333h;
With
sodium hydrogencarbonate;
In
1,2-dimethoxyethane; water;
Product distribution / selectivity;
|
72% |
|
With
lithium amide;
[(CyPF-tBu)PdCl2];
In
1,4-dioxane;
at 80 ℃;
for 24h;
Product distribution / selectivity;
|
37% |
|
With
lithium amide;
(R)-1-[(S)-2-(diphenylphosphino)ferrocenyl]ethyldi-t-butylphosphine; bis(dibenzylideneacetone)-palladium(0);
In
1,2-dimethoxyethane;
at 80 ℃;
for 24h;
Product distribution / selectivity;
|
|
|
With
lithium amide;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 80 ℃;
for 24h;
Product distribution / selectivity;
|
|
|
With
lithium amide;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 110 ℃;
for 6h;
Product distribution / selectivity;
|
|
|
With
lithium amide;
[(CyPF-tBu)PdCl2];
In
tetrahydrofuran;
at 80 ℃;
for 24h;
Product distribution / selectivity;
|
|
|
With
ammonia; sodium t-butanolate;
(R)-1-[(S)-2-(diphenylphosphino)ferrocenyl]ethyldi-t-butylphosphine; bis(dibenzylideneacetone)-palladium(0);
In
1,2-dimethoxyethane;
at 20 - 80 ℃;
under 4137.29 Torr;
Product distribution / selectivity;
Parr bomb;
|
|
|
With
ammonia; sodium t-butanolate;
[(CyPF-tBu)PdCl2];
In
toluene;
at 80 ℃;
for 6h;
Product distribution / selectivity;
Sealed tube;
|
|
|
With
ammonia; sodium t-butanolate;
[(CyPF-tBu)PdCl2];
In
1,4-dioxane;
at 80 ℃;
for 6h;
Product distribution / selectivity;
Sealed tube;
|
|
|
With
ammonia; sodium t-butanolate;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 20 - 90 ℃;
for 20 - 24h;
under 4137.29 Torr;
Product distribution / selectivity;
Sealed tube;
Parr bomb;
|
71 - 88 %Spectr. |
|
With
ammonia; sodium t-butanolate;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 80 ℃;
for 6 - 24h;
Product distribution / selectivity;
Sealed tube;
|
|
|
With
ammonia; sodium t-butanolate;
[(CyPF-tBu)PdCl2];
In
1,2-dimethoxyethane;
at 20 - 80 ℃;
under 4137.29 Torr;
Product distribution / selectivity;
Parr bomb;
|
-
-
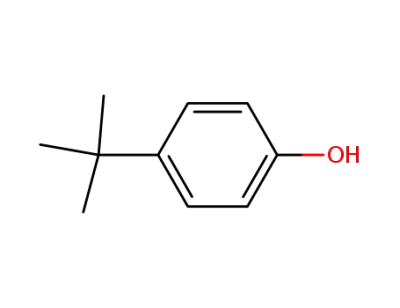
98-54-4
para-tert-butylphenol

-

-
4627-22-9
bis(4-tert-butylphenyl)amine

-

-
769-92-6
4-tert-Butylaniline
| Conditions | Yield |
|---|---|
|
With
ammonium bromide; bromo zinc ammonia;
at 320 - 330 ℃;
|
4627-22-9 Upstream products
-
98-54-4

para-tert-butylphenol
-
35779-04-5
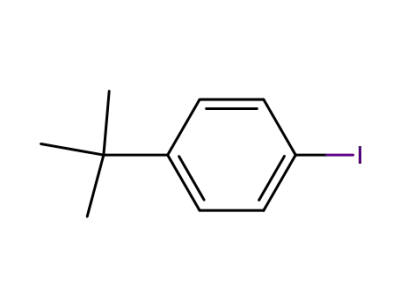
1-tert-butyl-4-iodobenzene
-
20330-45-4
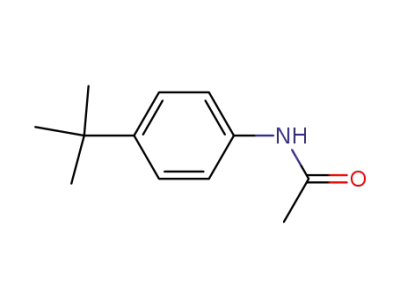
4-tert-butylacetanilide
-
769-92-6

4-tert-Butylaniline
4627-22-9 Downstream products
-
32367-71-8

Bis(4-tert.-butyl-phenyl)aminyl
-
134538-77-5
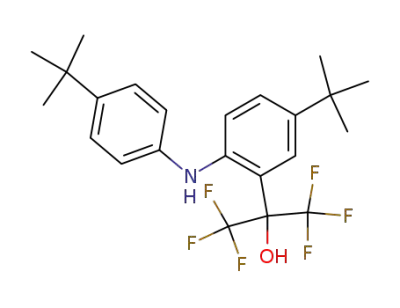
2-(1-hydroxy-1-trifluoromethyl-2,2,2-trifluoroethyl)-4,4'-di(tert-butyl)diphenylamine
-
134538-76-4
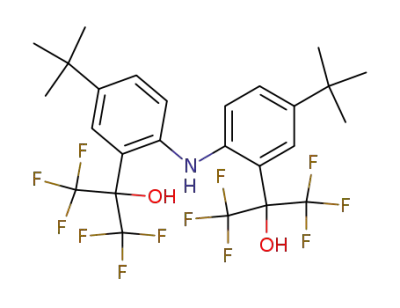
2,2'-di(1-hydroxy-1-trifluoromethyl-2,2,2-trifluoroethyl)-4,4'-di(tert-butyl)diphenylamine
-
98-54-4

para-tert-butylphenol
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
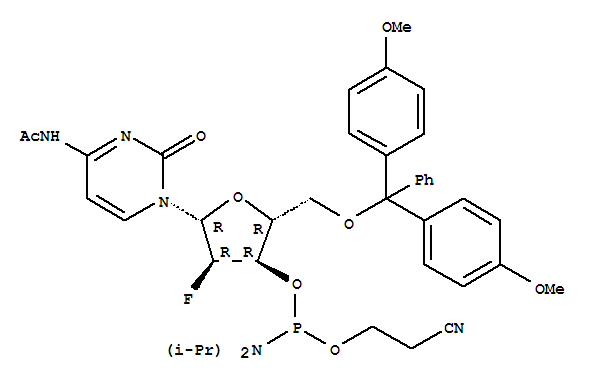
DMT-2’-F-dC(Ac)-CE-Phosphoramidite
CAS:159414-99-0
-
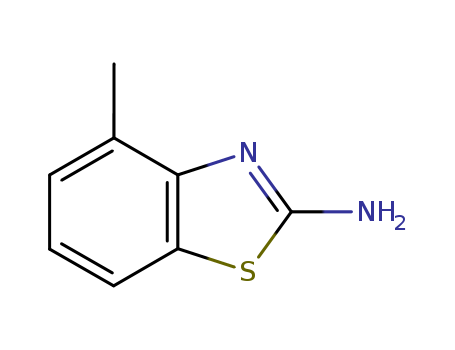
2-Amino-4-methylbenzothiazole
CAS:1477-42-5