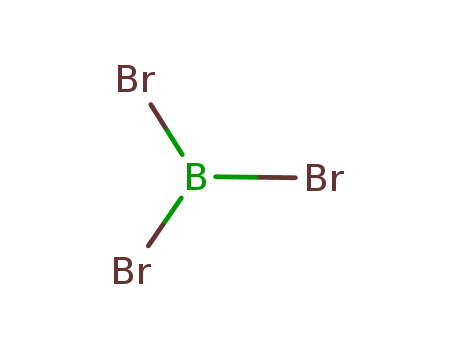
10294-33-4
- Product Name:Boron tribromide-11B
- Molecular Formula:BBr3
- Purity:99%
- Molecular Weight:250.523
Product Details;
CasNo: 10294-33-4
Molecular Formula: BBr3
Appearance: clear to amber liquid with a pungent odour
Bulk supply high purity Boron tribromide-11B 10294-33-4, Paid sample available
- Molecular Formula:BBr3
- Molecular Weight:250.523
- Appearance/Colour:clear to amber liquid with a pungent odour
- Vapor Pressure:40 mm Hg ( 14 °C)
- Melting Point:-46 °C(lit.)
- Refractive Index:1.549
- Boiling Point:91.3 °C at 760 mmHg
- Flash Point:30°F
- PSA:0.00000
- Density:2.782 g/cm3
- LogP:2.15600
Boron tribromide(Cas 10294-33-4) Usage
|
General Description |
Boron tribromide is commercially available and is a strong Lewis acid. It is an excellent demethylating or dealkylating agent for ethers, often in the production of pharmaceuticals. boron tribromide lewis structure Additionally, it also finds applications in olefin polymerisation and in Friedel–Crafts chemistry as a Lewis acid catalyst. The electronics industry uses boron tribromide as a boron source in pre-deposition processes for doping in the manufacture of semiconductors. Boron tribromide is a colourless, fuming liquid compound containing boron and bromine. It is usually made by heating boron trioxide with carbon in the presence of bromine: this generates free boron that reacts vigorously with the bromine. Boron tribromide is used extensively in industries associated with pharmaceutical manufacturing, image processing, semiconductor doping, plasma etching, and photovoltaic manufacturing and as a reagent for different chemical processes. |
|
Physical and Chemical Properties |
Fuming colorless viscous liquid with a strong irritant, toxic. Melting point is-46 ℃, the boiling point is 91.3 ℃. It was dissolved in carbon tetrachloride. Easily decomposed by water, alcohol. light or thermal decomposition, heated to explode. It can react with the phosphorus, nitrogen, oxygen, sulfur, halogens, ammonia, alkali, phosphorus halides, phosphines, and many substituents of ammonia. A strong corrosive. Strong irritative to Skin, eye or mucous membrane. Approximate toxicity of hydrogen bromide. United States provides operating maximum allowable concentration of boron tribromide in environment Air is 1ppm (10mg/m3). It is obtained in laboratory by Aluminum tribromide reacting with boron trifluoride, then distillation. can be used as a source of doped semiconductor silicon, but also for the preparation of high purity boron and organic boride. Other related chemical reactions involved by boron tribromide: In hydroiodic, at 300~400 ℃ continuously fed in boron tribromide, obtained mixture of BIBr2 and BI2Br, BI3, and then separated by distillation, derived dibromo iodide boron. Boron tribromide reacts with adamantine, generates 1-bromo-adamantane. The above information is edited by the lookchem of Yan Yanyong. |
|
Boron trichloride |
Boron trifluoride, boron trichloride, boron tribromide and boron triiodide are four kinds of common boron halides, the last three kinds of halogenated boron can be made in the presence of carbonaceous reducing agent by the high-temperature oxidation reduction of halogens and diboron trioxide, the reaction equation is as follows: B2O3 + 3C + 3Cl2 = 2BCl3 + 3CO, boron trifluoride is obtained by interaction of hydrogen fluoride (fluorspar with concentrated sulfuric acid) and diboron trioxide. Boron halide are all covalent molecules, in the vapor state existing in a planar triangles single molecule. Boron atoms using sp2 hybrid orbitals, p orbital of boron atoms filled of electron in the vertical plane perpendicular to the empty p orbital plane of a halogen atom can form large π bond π64. Experimental results show that the measured bond length (such as B-Cl bond length is 173pm) is shorter than a single bond (single bond B-Cl bond length is 187pm), indicating the presence of large π bond. The melting points of all these four kinds of halides are low, boron trifluoride is the lowest, and the boiling point increases with the increase of atomic number of halogen, indicating four kinds of halide are covalent halide molecules, intermolecular attraction is van der Waals forces. In 4 kinds of halides, stability is declined from boron trifluoride in turn to boron triiodide. Boron halides are easily hydrolyzed to produce boric acid. |
|
Toxicity |
Boron tribromide has a strong stimulating effect on human tissue, and its vapor is highly toxic, strong corrosive. Wear masks, gloves and protective clothing during operation. Steam inhalation is strictly prohibited. Immediately sent to hospital for treatment after poisoning. |
|
Production method |
Direct synthesis is putting the dried powder of boron into the reaction tube of a tubular reactor, to make the reaction can be carried out sufficiently, a certain amount of filler should be placed in the reaction tube, the filler material is the same as the inner wall of the reaction tube. The reaction tube was heated to 850 ℃, also bromine in the bromine vessel was heated to a boil, then poured into reaction tube. The boron tribromide liquid produced with activated carbon, zinc and aluminum scrap together in debromination vessel heated to reflux until boron bromide produced is a colorless, and then by crude distillation, distillation, obtained completely colorless bromide boron products. 2B + 3Br2 → 2BBr3 Salt Lake alkaline mineral general uses open-pit mining, ancient alkaline mineral general uses underground dissolution mining law. Mining process 1. open-pit mining 2. underground dissolution method |
|
Reactivity Profile |
Boron tribromide strongly attacks wood and rubber with generation of flammable hydrogen gas. Reacts exothermically and violently with water. Mixing tungsten trioxide and Boron tribromide caused an explosion when the reaction was not cooled in an ice bath. |
|
Hazard |
Corrosive to tissue. May explode when heated. Upper respiratory tract irritant. |
|
Health Hazard |
Inhalation causes severe irritation of mucous membranes. Ingestion causes burns of mouth and stomach. Contact with eyes or skin causes severe burns. |
|
Fire Hazard |
Special Hazards of Combustion Products: Toxic fumes of the chemical or hydrogen bromide may form in fires. |
|
Potential Exposure |
Boron tribromide is highly toxic and corrosive, it is used as a catalyst in organic synthesis, making diborane, high purity boron, and semiconductors. |
|
Shipping |
UN2692 Boron tribromide, Hazard class: 8; Labels: 8—Corrosive materials, 6.1—Poison Inhalation Hazard, Inhalation Hazard Zone B. |
|
Incompatibilities |
Reacts violently and explosively with water, steam, or alcohols, forming toxic, corrosive, and potentially explosive hydrogen bromide gas. Mixtures with potassium or sodium can explode on impact. Incompatible with oxidizers, strong bases. Attacks some metals, rubbers, and plastics. |
InChI:InChI=1/BBr3/c2-1(3)4
10294-33-4 Relevant articles
Fast H/D exchange of B, B′, B′-tribromoborazine in C 6D6 in the presence of aluminum tribromide: First evidence for an electrophilic substitution reaction of borazines in solution
Timoshkin, Alexey Y.,Kazakov, Igor V.,Lisovenko, Anna S.,Bodensteiner, Michael,Scheer, Manfred
, p. 9039 - 9044 (2011)
A solution of B,B′,B′-tribromoborazine (...
Hybrid 2-aminotetralin and aryl-substituted piperazine compounds and their use in altering cns activity
-
, (2008/06/13)
Hybrid compounds containing in aminotetr...
8-hydroxy-7-substituted quinolines as anti-viral agents
-
, (2008/06/13)
The present invention provides for 8-hyd...
METHOD FOR COUPLING LIVING CATIONIC POLYMERS
-
, (2008/06/13)
A method for coupling a living cationic ...
10294-33-4 Process route
-
-
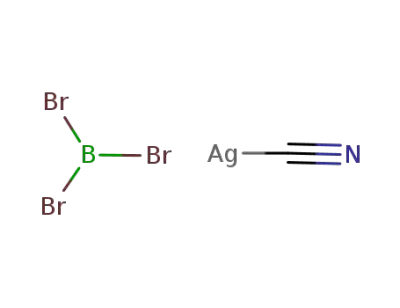
silver (I) cyanide * boron tribromide

-
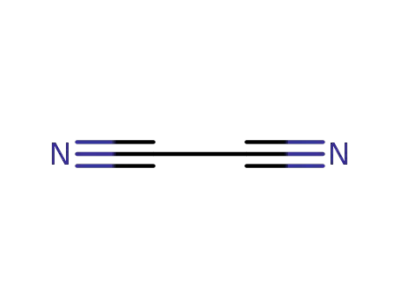
-
460-19-5,25215-76-3
ethanedinitrile

-
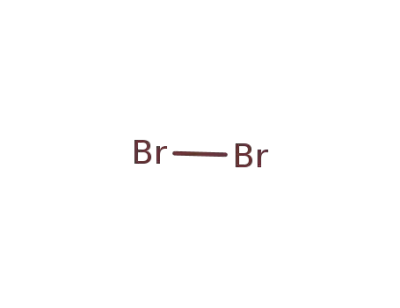
-
7726-95-6
bromine

-
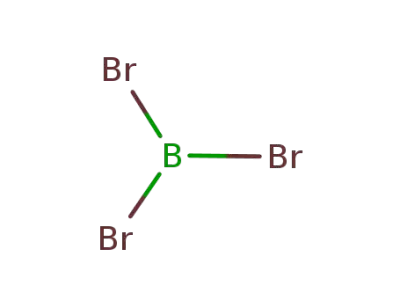
-
10294-33-4
boron tribromide
| Conditions | Yield |
|---|---|
|
In
neat (no solvent);
heating at 150°C;;
|
-
-
BBr3*Ag(1+)*CN(1-)=BBr3*AgCN

-
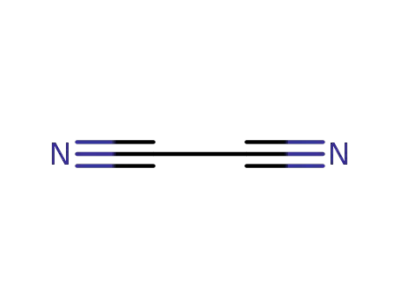
-
460-19-5,25215-76-3
ethanedinitrile

-
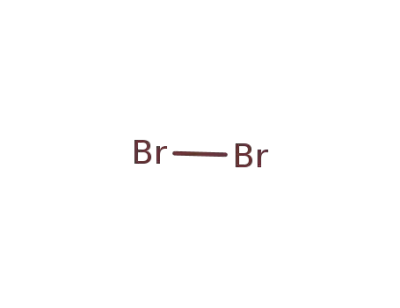
-
7726-95-6
bromine

-

-
10294-33-4
boron tribromide
| Conditions | Yield |
|---|---|
|
above 150°C;
|
10294-33-4 Upstream products
-
7726-95-6
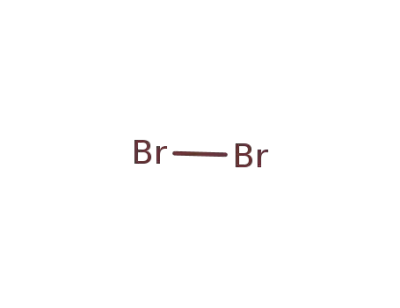
bromine
-
96-11-7
1,2,3-tribromopropane
-
13510-40-2
phosphorus trichloride dibromide
-
7440-44-0

pyrographite
10294-33-4 Downstream products
-
5123-17-1
bromodiphenylborane
-
259209-20-6
5-fluoro-2-hydroxyphenyl-boronic acid
-
212755-09-4
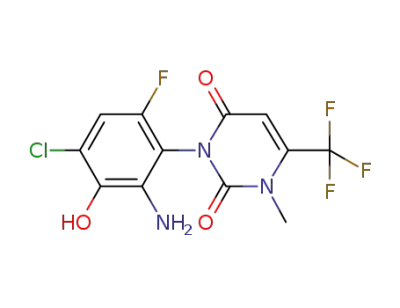
1-methyl-6-trifluoromethyl-3-(6-amino-4-chloro-2-fluoro-5-hydroxy-phenyl)-2,4(1H,3H)-pyrimidinedione
-
199676-41-0
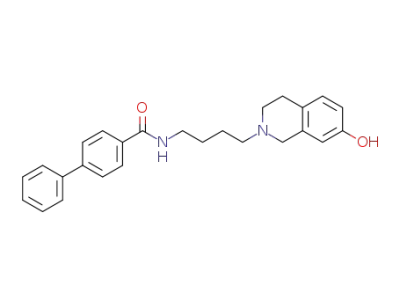
7-hydroxy-N-(4-(4-phenylbenzoylamino)butyl)-1,2,3,4-tetrahydroisoquinoline
Relevant Products
-
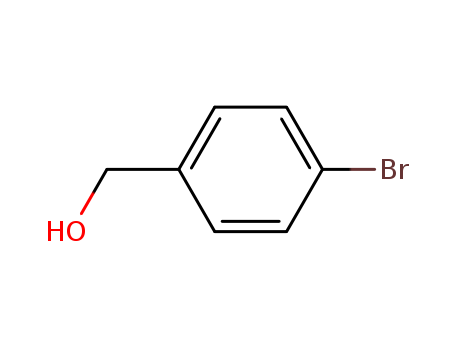
4-Bromobenzyl alcohol
CAS:873-75-6
-
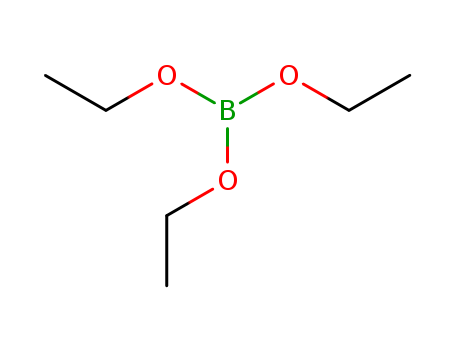
Triethyl borate-11B
CAS:150-46-9
-
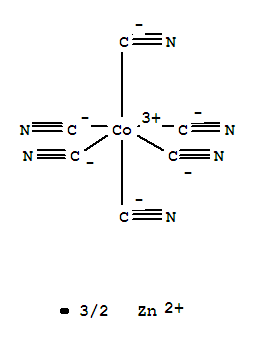
Zinchexacyanocobaltate
CAS:14049-79-7