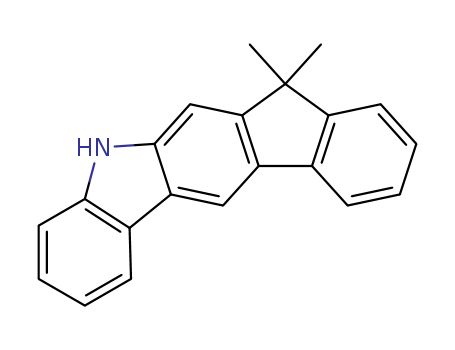
56-87-1
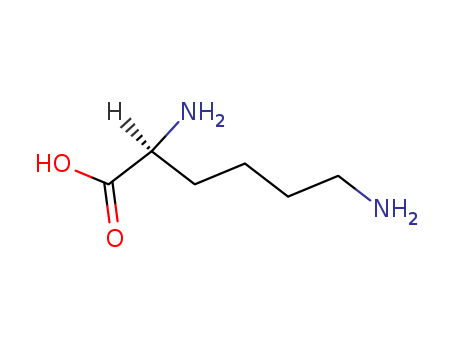
- Product Name:L-Lysine
- Molecular Formula:C6H14N2O2
- Purity:99%
- Molecular Weight:146.189
Product Details;
CasNo: 56-87-1
Molecular Formula: C6H14N2O2
Appearance: white or almost white crystalline powder
Factory Supply industrial standard L-Lysine 56-87-1 In Stock
- Molecular Formula:C6H14N2O2
- Molecular Weight:146.189
- Appearance/Colour:white or almost white crystalline powder
- Melting Point:215 °C (dec.)(lit.)
- Refractive Index:26 ° (C=2, 5mol/L HCl)
- Boiling Point:311.542 °C at 760 mmHg
- Flash Point:142.216 °C
- PSA:89.34000
- Density:1.125 g/cm3
- LogP:0.92790
Lysine(Cas 56-87-1) Usage
|
Essential Amino Acid |
Lysine is an indispensable amino acid (IAA) and is essential in our diets, meaning it must be obtained from food as the body cannot synthesize it. Its importance was demonstrated by William Rose and colleagues in 1955, showing that lysine is required in the human diet for positive nitrogen balance. |
|
Amino Acid Score (AAS) |
Lysine is singled out as an important IAA because it is present in limited amounts in many food sources, particularly grains. The FAO/WHO has established an amino acid score based on the ideal composition of each IAA in protein. Lysine concentration in protein is compared to an ideal protein, and its AAS should ideally be 45 mg/g protein for adults. |
|
Protein Synthesis |
Lysine plays a crucial role in protein synthesis. Proteins cannot be synthesized if any essential amino acid, like lysine, is limited in availability. Limiting lysine intake results in reduced protein synthesis and can lead to a negative nitrogen balance. |
|
Lysine Deficiency |
Foods like grains and peanut butter have lysine concentrations below the ideal level, resulting in AAS scores below 1. Lysine deficiency can be overcome by consuming larger amounts of protein or by combining lysine-deficient foods with lysine-rich foods, such as beans, to achieve a balanced amino acid intake. |
|
Role in Animals |
Lysine cannot be synthesized by mammals and is therefore essential in their diets as well. Lysine is strictly indispensable and does not participate in transamination reactions. It serves as a precursor for protein synthesis and is catabolized through the saccharopine pathway, primarily in the liver. |
|
Biosynthesis of Carnitine |
Lysine is also a precursor for the biosynthesis of carnitine, an important compound involved in fatty acid metabolism. Carnitine plays a crucial role in transporting fatty acids into the mitochondria for 尾-oxidation, contributing to energy production. |
|
General Description |
Lysine is an essential amino acid involved in various biological processes, including protein synthesis, enzyme activity modulation, and the biosynthesis of important compounds like pipecolic acid in natural products such as rapamycin. It serves as a precursor for homologues like (S)-homolysine, which are valuable in drug discovery, particularly for peptidic enzyme inhibitors. Lysine also plays a role in glycation reactions, forming Maillard products that influence intestinal absorption and transport mechanisms. Additionally, its derivatives, such as lysine-dopamine (LDA), are utilized to enhance surface biocompatibility for biomedical applications. Its versatility extends to synthetic chemistry, where it is incorporated into peptide motifs for bioactive compound development. |
InChI:InChI=1/C6H14N2O2/c7-4-2-1-3-5(8)6(9)10/h5H,1-4,7-8H2,(H,9,10)/t5-/m0/s1
56-87-1 Relevant articles
Binding Methylarginines and Methyllysines as Free Amino Acids: A Comparative Study of Multiple Host Classes**
Warmerdam, Zoey,Kamba, Bianca E.,Le, My-Hue,Schrader, Thomas,Isaacs, Lyle,Bayer, Peter,Hof, Fraser
, (2021/11/30)
Methylated free amino acids are an impor...
DISCOVERY, TOTAL SYNTHESIS, AND BIOACTIVITY OF DOSCADENAMIDES
-
Page/Page column 81; 84-85, (2021/02/05)
The invention is directed towards compou...
Direct monitoring of biocatalytic deacetylation of amino acid substrates by1H NMR reveals fine details of substrate specificity
De Cesare, Silvia,McKenna, Catherine A.,Mulholland, Nicholas,Murray, Lorna,Bella, Juraj,Campopiano, Dominic J.
supporting information, p. 4904 - 4909 (2021/06/16)
Amino acids are key synthetic building b...
Development of a Raltegravir-based Photoaffinity-Labeled Probe for Human Immunodeficiency Virus-1 Integrase Capture
Pala, Nicolino,Esposito, Francesca,Tramontano, Enzo,Singh, Pankaj Kumar,Sanna, Vanna,Carcelli, Mauro,Haigh, Lisa D.,Satta, Sandro,Sechi, Mario
supporting information, p. 1986 - 1992 (2020/11/09)
Photoaffinity labeling (PAL) is one of t...
56-87-1 Process route
-
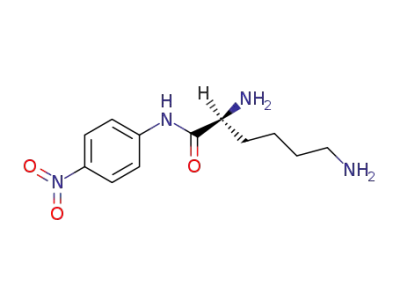
-
19826-45-0,6184-11-8
L-lysine-p-nitroanilide

-
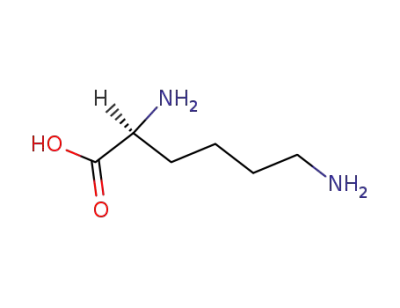
-
56-87-1,3506-25-0
L-lysine

-
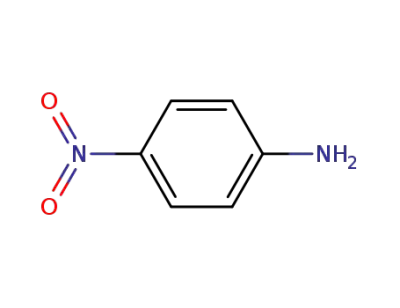
-
100-01-6,104810-17-5
4-nitro-aniline
| Conditions | Yield |
|---|---|
|
With
Streptomyces griseus dizinc aminopeptidase; water; calcium(II) ion;
In
various solvent(s);
at 30 ℃;
pH=8.0;
Enzyme kinetics;
|
|
|
With
Tris-HCl buffer; an aminopeptidase from the seeds of Cannabis sativa; water;
at 37 ℃;
pH=7.5;
Enzyme kinetics;
Enzymatic reaction;
|
-
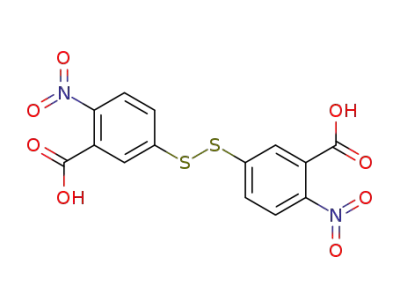
-
69-78-3
5,5'-dithiobis-(2-nitrobenzoic acid)

-
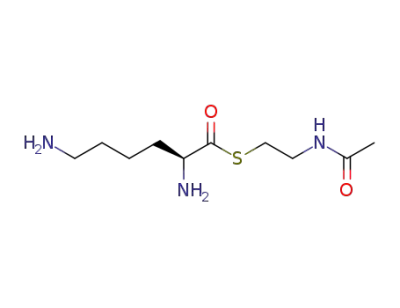
-
L-Lys-S-(N-acetyl)cysteamine

-

-
56-87-1,3506-25-0
L-lysine

-
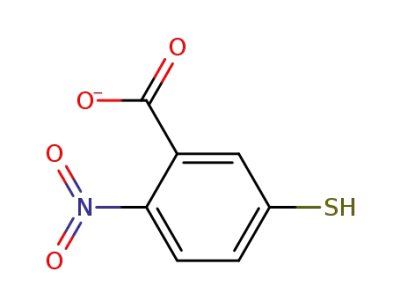
-
18430-02-9,39509-22-3
2-nitro-5-mercaptobenzoic acid anion

-
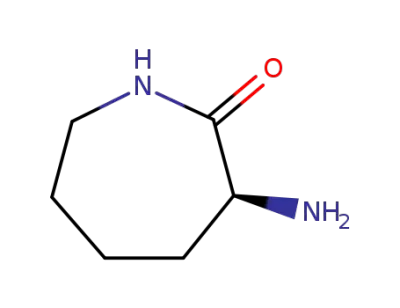
-
21568-87-6
2-aminocaprolactam
| Conditions | Yield |
|---|---|
|
In
aq. buffer;
at 30 ℃;
pH=7.5;
Kinetics;
|
56-87-1 Upstream products
-
2100-17-6
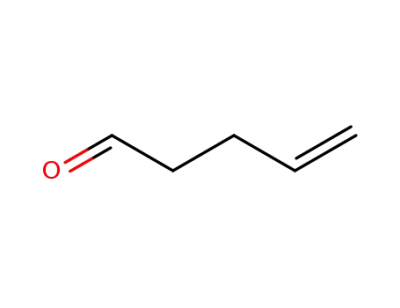
4-pentenal
-
627-76-9
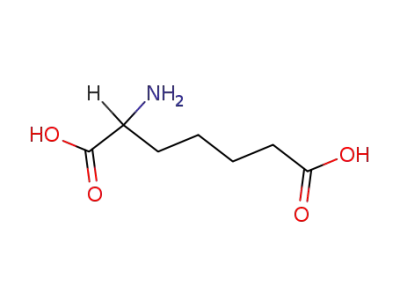
alpha-aminopimelic acid
-
10508-34-6
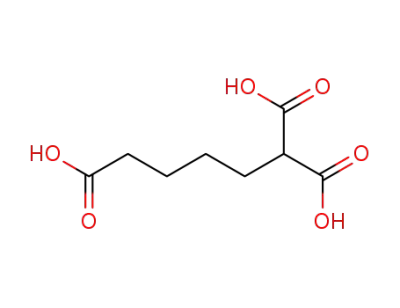
pentane-1,1,5-tricarboxylic acid
-
71264-11-4
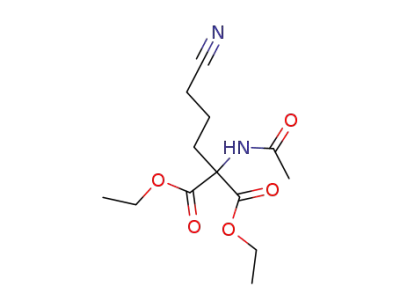
diethyl 2-acetamido-2-(3-cyanopropyl)malonate
56-87-1 Downstream products
-
56-87-1

L-lysine
-
156-86-5
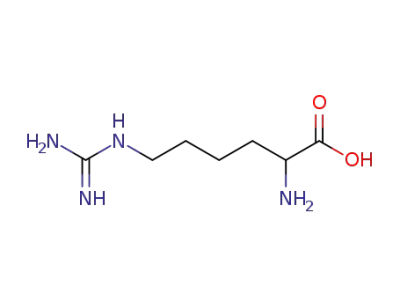
homoarginine
-
2756-89-0
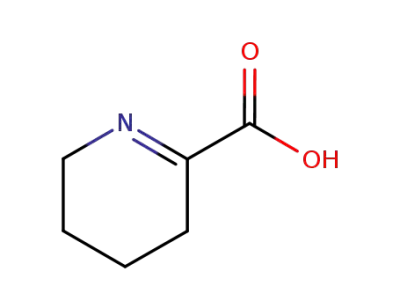
Δ1-piperidine-2-carboxylic acid
-
923-27-3
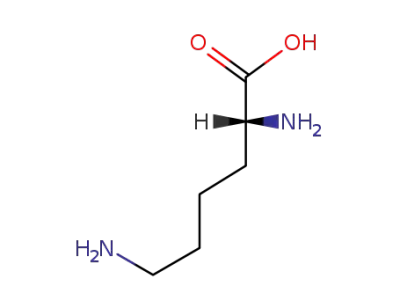
D-lysine
Relevant Products
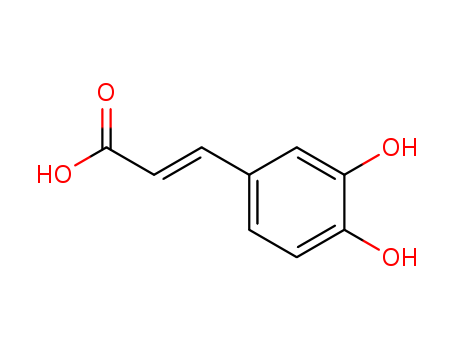
-
benfooxythiamine
CAS:909542-99-0
-
5,7-Dihydro-7,7-dimethyl-indeno[2,1-b]carbazole
CAS:1257220-47-5
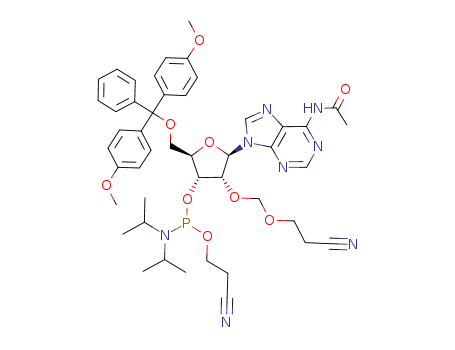
-
2’OCEM-rA(Ac) phosphoramidite
CAS:863408-46-2