2426-08-6
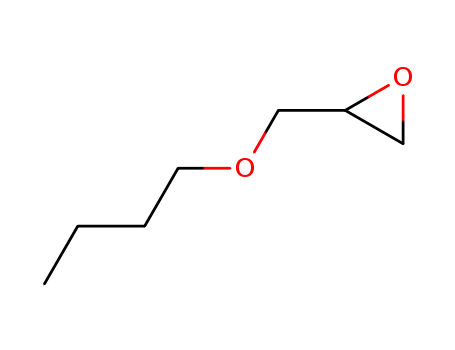
- Product Name:BUTYL GLYCIDYL ETHER
- Molecular Formula:C7H14O2
- Purity:99%
- Molecular Weight:130.187
Product Details;
CasNo: 2426-08-6
Molecular Formula: C7H14O2
Appearance: colourless liquid
Factory sells BUTYL GLYCIDYL ETHER 2426-08-6 with sufficient production capacity
- Molecular Formula:C7H14O2
- Molecular Weight:130.187
- Appearance/Colour:colourless liquid
- Vapor Pressure:1.43mmHg at 25°C
- Melting Point:59 °C
- Refractive Index:n20/D 1.418(lit.)
- Boiling Point:176.999 °C at 760 mmHg
- Flash Point:54.444 °C
- PSA:21.76000
- Density:0.945 g/cm3
- LogP:1.20190
Butyl glycidyl ether(Cas 2426-08-6) Usage
|
Production Methods |
n-Butyl glycidyl ether (BGE) is made by the condensation of n-butyl alcohol and epichlorohydrin with subsequent dehydrochlorination with caustic to form the epoxy ring. |
|
Air & Water Reactions |
Highly flammable. Butyl glycidyl ether may be sensitive to prolonged exposure to air, may form explosive peroxide in contact with air. |
|
Reactivity Profile |
Butyl glycidyl ether, an ether, can act as a base. They form salts with strong acids and addition complexes with Lewis acids. The complex between diethyl ether and boron trifluoride is an example. Ethers may react violently with strong oxidizing agents. In other reactions, which typically involve the breaking of the carbon-oxygen bond, ethers are relatively inert. |
|
Hazard |
A mild skin and eye irritant. Sensitization and reproduction effects. |
|
Health Hazard |
Exposure can cause mild irritation of skin, eyes, nose, and respiratory tract. Chronic exposure may cause inflammation and sensitization of the skin. |
|
Flammability and Explosibility |
Flammable |
|
Contact allergens |
A reactive diluent used to reduce viscosity of epoxy resins Bisphenol A type. |
|
Safety Profile |
Suspected Carcinogen. Moderately toxic by ingestion, skin contact, and inuaperitoneal routes. Mildly toxic by inhalation. An experimental teratogen. Mutation data reported. A sktn and severe eye irritant. See also ETHERS. When heated to decomposition it emits acrid and irritating fumes. |
|
Potential Exposure |
NIOSH has estimated human exposures @ 18,000. Used as reactive diluent for epoxy resins, flooring, laminating, and electrical; and as a stabilizer, viscosity-reducing agent, as acid acceptor for solvents; and as a chemical intermediate |
|
Carcinogenicity |
BGE was mutagenic in bacterial assays, and DNA damage was induced in human cells in vitro. |
|
Shipping |
UN1993 Flammable liquids, n.o.s., Hazard Class: 3; Labels: 3—Flammable liquid, Technical Name Required |
|
Incompatibilities |
May form explosive mixture with air. Air and light form unstable and explosive peroxides. Contact with strong oxidizers may cause fire and explosions. Contact with strong caustics may cause polymerization. Attacks some plastics and rubber |
|
Waste Disposal |
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed. |
|
General Description |
Colorless to pale yellow liquid with a strong, slightly unpleasant odor. Flash point approximately 164°F. Denser than water. Vapors are heavier than air. Vapors may irritate the nose, throat and respiratory tract. Ingestion or inhalation may cause central nervous system depression. Liquid contact may severely irritate the eyes and skin. Prolonged contact with the skin may cause defatting and drying. |
InChI:InChI=1/C7H14O2/c1-2-3-4-8-5-7-6-9-7/h7H,2-6H2,1H3/t7-/m0/s1
2426-08-6 Relevant articles
An Amphiphilic (salen)Co Complex – Utilizing Hydrophobic Interactions to Enhance the Efficiency of a Cooperative Catalyst
Solís-Mu?ana, Pablo,Salam, Joanne,Ren, Chloe Z.-J.,Carr, Bronte,Whitten, Andrew E.,Warr, Gregory G.,Chen, Jack L.-Y.
supporting information, p. 3207 - 3213 (2021/06/01)
An amphiphilic (salen)Co(III) complex is...
Chiral amorphous metal–organic polyhedra used as the stationary phase for high-resolution gas chromatography separations
Tang, Bo,Sun, Chenyu,Wang, Wei,Geng, Lina,Sun, Liquan,Luo, Aiqin
, p. 1178 - 1185 (2020/07/09)
Herein, we describe a new chiral amorpho...
Diastereoselective Desymmetrization of p-Quinamines through Regioselective Ring Opening of Epoxides and Aziridines
Jadhav, Sandip B.,Chegondi, Rambabu
supporting information, p. 10115 - 10119 (2019/12/24)
A highly diastereoselective desymmetriza...
Asymmetric Hydrolytic and Aminolytic Kinetic Resolution of Racemic Epoxides using Recyclable Macrocyclic Chiral Cobalt(III) Salen Complexes
Tak, Rajkumar,Kumar, Manish,Menapara, Tusharkumar,Gupta, Naveen,Kureshy, Rukhsana I.,Khan, Noor-ul H.,Suresh
supporting information, p. 3990 - 4001 (2017/11/22)
New chiral macrocyclic cobalt(III) salen...
2426-08-6 Process route
-
-
2426-08-6
glycidyl n-butyl ether

-
-
98451-40-2,119066-32-9,119066-33-0,624-52-2
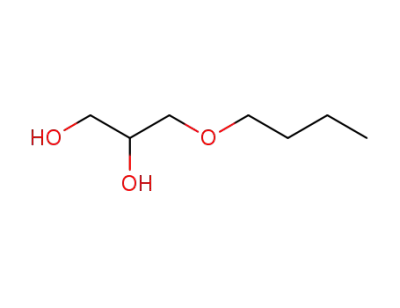
3-n-butoxy-1,2-propanediol

-
-
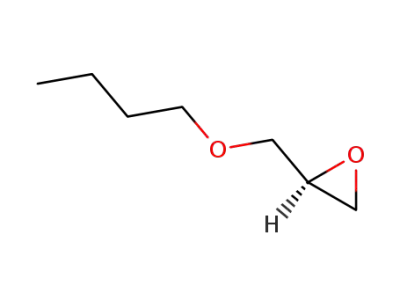
2426-08-6,85858-60-2,121906-41-0,130232-96-1
(R)-3-(Butoxy)-1,2-epoxypropane
| Conditions | Yield |
|---|---|
|
glycidyl n-butyl ether;
With
C57H54CoN3O8;
In
dichloromethane;
at 20 ℃;
for 0.25h;
With
water;
In
dichloromethane;
at 20 ℃;
for 12h;
enantioselective reaction;
Cooling with ice;
|
44% |
|
With
water; C71H111CoN2O9S;
In
neat (no solvent);
at 20 ℃;
for 0.416667h;
enantioselective reaction;
Resolution of racemate;
Inert atmosphere;
Schlenk technique;
|
> 99 % ee |
-

-
2426-08-6
glycidyl n-butyl ether

-

-
98451-40-2,119066-32-9,119066-33-0,624-52-2
3-n-butoxy-1,2-propanediol

-

-
1436-34-6,104898-06-8,122922-40-1,130404-08-9
(S)-1,2-epoxyhexane

-

-
2426-08-6,85858-60-2,121906-41-0,130232-96-1
(R)-3-(Butoxy)-1,2-epoxypropane
| Conditions | Yield |
|---|---|
|
With
(R,R)-Salen-Co-OTs; water;
In
neat (no solvent);
at 20 ℃;
for 0.416667h;
enantioselective reaction;
Resolution of racemate;
Inert atmosphere;
Schlenk technique;
|
67 % ee |
2426-08-6 Upstream products
-
106-89-8
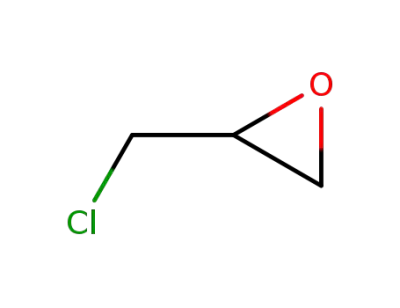
epichlorohydrin
-
98451-40-2

3-n-butoxy-1,2-propanediol
-
16224-33-2
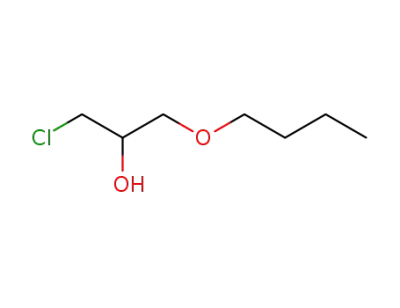
1-chloro-3-n-butoxy-2-propanol
-
3739-64-8
allyl n-butyl ether
2426-08-6 Downstream products
-
861055-29-0
acetic acid-(3-butoxy-2-hydroxy-propylamide)
-
32972-04-6
1-diethylamino-3-butoxy-2-propanole
-
17539-86-5
5-butoxymethyl-3-p-tolyl-oxazolidin-2-one
-
26798-85-6
C27H47Cl3O6Si
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
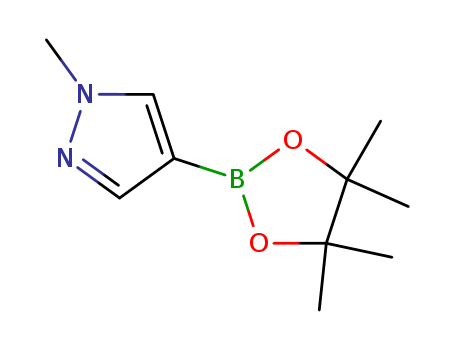
1-Methyl-4-pyrazole boronic acid pinacol ester
CAS:761446-44-0
-
CHITOSAN 90%
CAS:9012-76-4