118-12-7
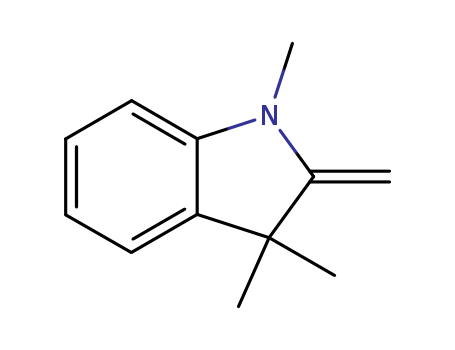
- Product Name:2-Methylene-1,3,3-trimethylindoline
- Molecular Formula:C12H15N
- Purity:99%
- Molecular Weight:173.258
Product Details;
CasNo: 118-12-7
Molecular Formula: C12H15N
Appearance: clear red liquid
Top Quality 2-Methylene-1,3,3-trimethylindoline 118-12-7 Hot Sell In Stock
- Molecular Formula:C12H15N
- Molecular Weight:173.258
- Appearance/Colour:clear red liquid
- Vapor Pressure:0.0249mmHg at 25°C
- Melting Point:-8 °C
- Refractive Index:n20/D 1.577(lit.)
- Boiling Point:248 °C at 760 mmHg
- PKA:2.19±0.40(Predicted)
- Flash Point:101.7 °C
- PSA:3.24000
- Density:1 g/cm3
- LogP:2.99270
1,3,3-Trimethyl-2-methyleneindoline(Cas 118-12-7) Usage
|
General Description |
1,3,3-Trimethyl-2-methyleneindoline (Fischer base) is a versatile intermediate in organic synthesis, particularly for introducing ethynyl groups into aryl and heteroaryl compounds via its acyl derivatives. It reacts with acylating agents and phosphorus oxychloride to form indolenium salts, which, upon treatment with aqueous alkali, yield monosubstituted acetylenes and 1,3,3-trimethyl-2-oxindole. This method is effective for aromatic and heterocyclic systems but unsuitable for alkylacetylenes due to resinification. The Fischer base thus serves as a key building block for synthesizing acetylenes, which are valuable in producing dyes and thermochromic materials. |
InChI:InChI=1/C12H15N/c1-9-12(2,3)10-7-5-6-8-11(10)13(9)4/h5-8H,1H2,2-4H3
118-12-7 Relevant articles
A NEW METHOD OF SYNTHESIS OF CONJUGATED SYSTEMS WITH TRIS(FLUOROMETHINE) CHAIN
Pazenok, S. V.,Kovtyukh, I. P.,Yagupolskii, L. M.
, p. 4595 - 4598 (1991)
The interaction of compounds having the ...
Synthesis of spiropyran with methacrylate at the benzopyran moiety and control of the water repellency and cell adhesion of its polymer film
Ajiro, Hiroharu,Ando, Tsuyoshi,Murase, Nobuo
, p. 1489 - 1495 (2020)
Stimuli-responsive materials have been a...
Crystal and solution structures of photochromic spirobenzothiopyran. First full characterization of the meta-stable colored species
Hirano, Masafumi,Osakada, Kohtaro,Nohira, Hiroyuki,Miyashita, Akira
, p. 533 - 540 (2002)
Full elucidation for stable, colorless, ...
Interaction studies between photochromic spiropyrans and transition metal cations: The curious case of copper
Natali, Manuel,Giordani, Silvia
, p. 1162 - 1171 (2012)
A series of four spiropyrans bearing dif...
Structures of Lithium Salts of 2,3,3-Trimethylindolenine and Its 5-Methoxy Derivative in Solution and the Solid State
Jackman, L. M.,Scarmoutzos, L. M.,Smith, B. D.,Williard, Paul G.
, p. 6058 - 6063 (1988)
An X-ray crystal structure of lithium 2,...
Unusual thermo(photo)chromic properties of some mononitro- and dinitro- substituted 3′-alkyl indolospirobenzopyrans This paper is dedicated to the memory of Dr Thomas G. Nevell.
Abdullah, Ayse,Nevell, Thomas G.,Sammes, Peter G.,Roxburgh, Craig J.
, p. 57 - 72 (2015)
Isomeric equilbria of dinitro-substitute...
-
Bernauer et al.
, p. 717,723 (1960)
-
NOVEL SQUARYLIUM COMPOUNDS, AND COMPOSITIONS COMPRISING SAME
-
Paragraph 0107; 0113-0115, (2021/04/29)
The present disclosure is disclosed here...
Methylation synthesis method of N-heterocyclic compound
-
Paragraph 0013, (2021/02/06)
The invention relates to a methylation s...
Method for preparing indoline
-
Paragraph 0016; 0018; 0019; 0021; 0024, (2021/01/12)
The invention relates to a method for pr...
118-12-7 Process route
-
-
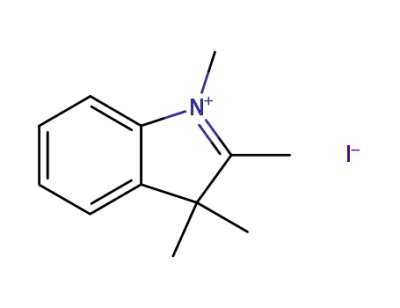
5418-63-3
1,2,3,3-tetramethyl-3H-indolium iodide

-
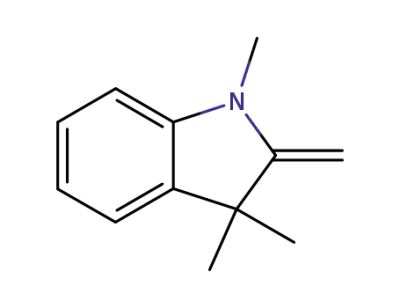
-
118-12-7
1,3,3-Trimethyl-2-methyleneindoline
| Conditions | Yield |
|---|---|
|
With
sodium hydroxide;
In
water;
at 20 ℃;
for 4h;
|
90% |
|
With
sodium hydroxide;
In
diethyl ether; water;
for 0.5h;
|
71% |
|
With
potassium hydroxide;
at 20 ℃;
for 1h;
|
55% |
|
With
potassium hydroxide;
Ambient temperature;
|
|
|
With
sodium hydroxide;
|
|
|
With
potassium hydroxide;
In
water; toluene;
|
|
|
With
sodium hydroxide;
at 20 ℃;
|
|
|
With
sodium hydroxide;
In
diethyl ether;
|
|
|
Multi-step reaction with 2 steps
1: durch Destillation im Kohlensaeurestrom oder im Vakuum
2: 100 °C
|
|
|
With
potassium hydroxide;
|
|
|
With
sodium hydroxide;
In
water; acetone;
|
|
|
With
sodium carbonate;
In
water;
at 20 ℃;
|
|
|
With
sodium hydroxide;
In
water;
|
|
|
With
sodium carbonate;
In
water;
at 20 ℃;
for 0.0166667h;
|
|
|
With
sodium hydroxide;
In
diethyl ether;
for 0.5h;
|
|
|
With
triethylamine;
In
ethanol;
at 70 ℃;
for 1h;
|
|
|
With
sodium hydroxide;
at 20 ℃;
for 0.166667h;
|
|
|
With
piperidine;
In
butanone;
for 0.0833333h;
Reflux;
Inert atmosphere;
|
-
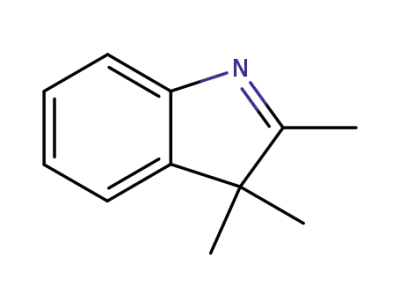
-
1640-39-7
2,3,3-trimethylindoleniune

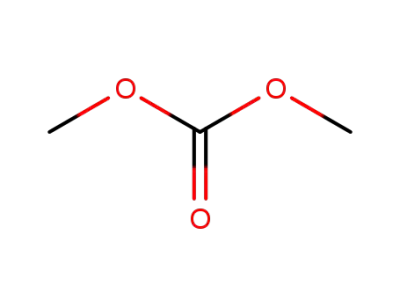
-
-
616-38-6
carbonic acid dimethyl ester

-

-
118-12-7
1,3,3-Trimethyl-2-methyleneindoline
| Conditions | Yield |
|---|---|
|
at 180 - 220 ℃;
under 22502.3 - 37503.8 Torr;
Inert atmosphere;
Sealed tube;
Autoclave;
|
97.8% |
118-12-7 Upstream products
-
120-72-9
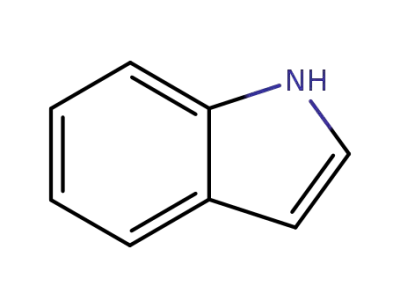
indole
-
74-88-4

methyl iodide
-
95-20-5
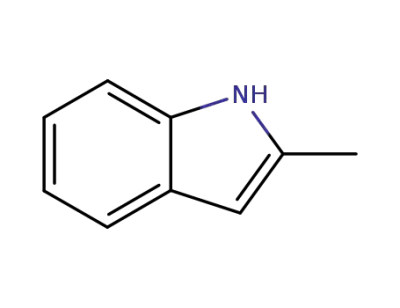
2-methyl-1H-indole
-
67-56-1
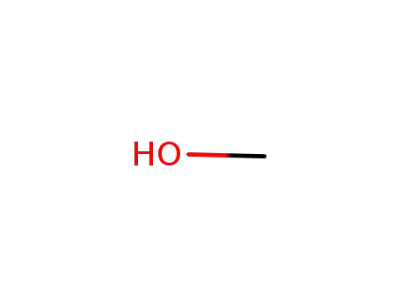
methanol
118-12-7 Downstream products
-
97301-42-3
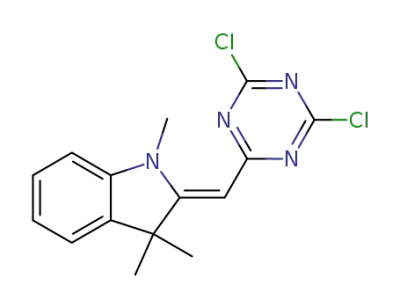
<1,3,3-Trimethyl-indolinyliden-(2)>-<4,6-dichlor-1,3,5-triazinyl-(2)>-methan
-
93872-94-7
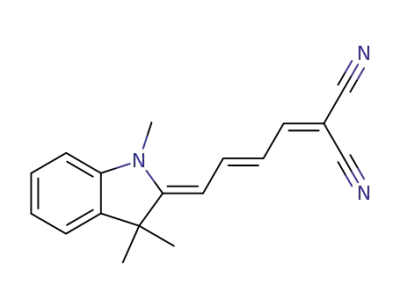
[4-(1,3,3-trimethyl-indolin-2-ylidene)-but-2-enylidene]-malononitrile
-
74145-97-4
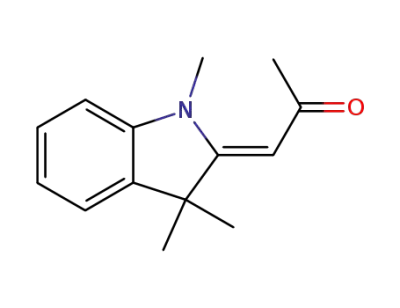
2-acetylmethylene-1,3,3-trimethylindoline
-
24293-93-4
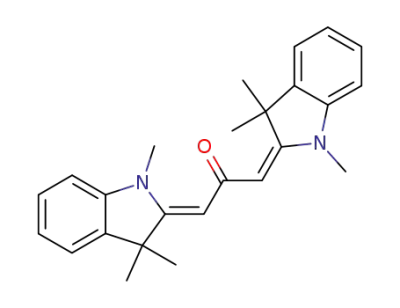
Bis-<1.3.3-trimethyl-indolinyliden-(2)-methyl>-keton
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
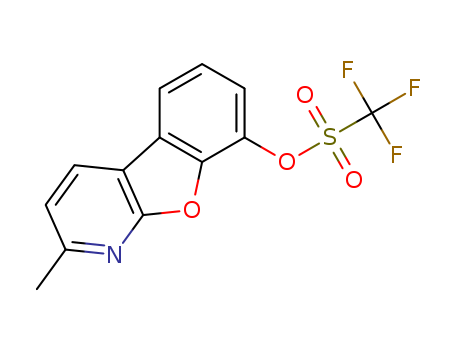
2-Methylbenzofuro[2,3-b]pyridin-8-yl trifluoromethanesulfonate
CAS:1609373-98-9
-
8-chloro-benzo[b]naphtho[2,1-d]furan
CAS:2103931-84-4