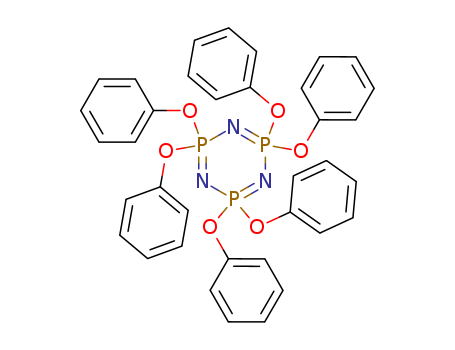
1184-10-7
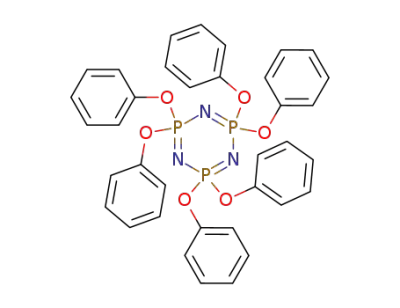
- Product Name:hexaphenoxycyclotriphosphazene
- Molecular Formula:C36H30N3O6P3
- Purity:99%
- Molecular Weight:693.572
Product Details;
CasNo: 1184-10-7
Molecular Formula: C36H30N3O6P3
China cas 1184-10-7 manufacturer wholesale hexaphenoxycyclotriphosphazene at affordable price
- Molecular Formula:C36H30N3O6P3
- Molecular Weight:693.572
- Vapor Pressure:0mmHg at 25°C
- Melting Point:116℃
- Boiling Point:696.518°C at 760 mmHg
- PKA:-12.47±0.50(Predicted)
- Flash Point:375.041°C
- PSA:121.89000
- Density:1.31 g/cm3
- LogP:10.61940
Phenoxycycloposphazene(Cas 1184-10-7) Usage
InChI:InChI=1/C36H30N3O6P3/c1-7-19-31(20-8-1)40-38-46(42-33-23-11-3-12-24-33)37-48(44-35-27-15-5-16-28-35,45-36-29-17-6-18-30-36)39(41-32-21-9-2-10-22-32)47(38)43-34-25-13-4-14-26-34/h1-30H
1184-10-7 Relevant articles
Synthesis and flame retardant properties of low density polyethylene/ethylene-vinyl acetate/polyphosphazene derivative composites
Zhao, Zhengping,Zhou, Zeping,Yu, Fengying,Ji, Jianbing
, p. 919 - 924 (2015)
Polyphosphazene derivative, hexaphenoxyl...
Multiporphyrin arrays on cyclotriphosphazene scaffolds
Kaur, Tejinder,Rajeswararao, Malakalapalli,Ravikanth, Mangalampalli
, p. 11051 - 11059 (2014)
We report the synthesis of first example...
Microwave-assisted synthesis of hexasubstituted cyclotriphosphazenes
Ye, Chengfeng,Liu, Weimin,Chen, Yunxia,Ou, Zhongwen
, p. 376 - 379 (2001)
-
Photophysical studies on multichromophoric cyclotriphosphazenes. Trinuclear excimer formation in hexakis(2-naphthyloxy)cyclotriphosphazene
Chattopadhyay, Nitin,Haldar, Basudeb,Mallick, Arabinda,Sengupta, Saumitra
, p. 3089 - 3092 (2005)
Hexakis(2-naphthyloxy)cyclotriphosphazen...
Fluorescence properties of fluorenylidene bridged cyclotriphosphazenes bearing aryloxy groups
?ift?i, G?nül Yenilmez,?enkuytu, Elif,Incir, Elif Saadet,Durmu?, Mahmut,Yuksel, Fatma
, p. 741 - 749 (2015)
The synthesis and characterization of th...
Synthesis method of hexaphenoxy cyclotriphosphazene flame retardant
-
Paragraph 0024-0039, (2021/06/26)
The invention discloses a synthesis meth...
Phosphazene functionalized silsesquioxane-based porous polymers for absorbing I2, CO2 and dyes
Wang, Yiqi,Soldatov, Mikhail,Wang, Qingzheng,Liu, Hongzhi
, (2021/03/06)
Porous polymers have been widely used as...
Method for synthesizing phenoxycycloposphazene
-
Paragraph 0022-0034, (2020/02/20)
The invention discloses a method for syn...
Tris(o-phenylenedioxy)cyclotriphosphazene as a Promoter for the Formation of Amide Bonds between Aromatic Acids and Amines
Movahed, Farzaneh Soleymani,Sawant, Dinesh N.,Bagal, Dattatraya B.,Saito, Susumu
, p. 3253 - 3262 (2020/11/02)
The atom-efficient formation of amide bo...
1184-10-7 Process route
-
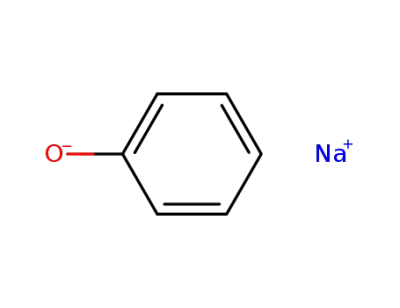
-
139-02-6
sodium phenoxide

-
-
1184-10-7
2,2,4,4,6,6-hexaphenoxycyclotriphosphazene
| Conditions | Yield |
|---|---|
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
acetonitrile;
at 30 - 80 ℃;
for 5h;
|
95.24% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
|
|
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
1,4-dioxane; diethyl ether;
Heating;
|
|
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
tetrahydrofuran;
Inert atmosphere;
|
-
-
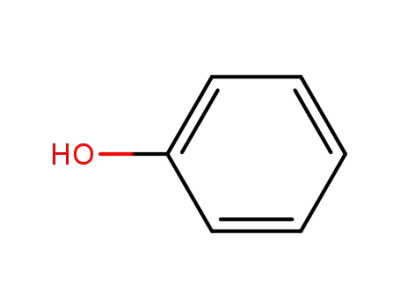
108-95-2,27073-41-2
phenol

-

-
1184-10-7
2,2,4,4,6,6-hexaphenoxycyclotriphosphazene
| Conditions | Yield |
|---|---|
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; potassium hydroxide;
In
acetonitrile;
at 80 ℃;
for 2h;
Reagent/catalyst;
Temperature;
Solvent;
Inert atmosphere;
|
99% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 1 - 4h;
Product distribution / selectivity;
|
98.1% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
chlorobenzene;
at 140 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)3ZnCl5;
In
chlorobenzene;
at 20 - 140 ℃;
for 5h;
Product distribution / selectivity;
Heating / reflux;
|
98.4% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
ammonium chloride; zinc(II) oxide;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 1.5 - 2.5h;
Product distribution / selectivity;
|
98% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
cobalt(II) chloride;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 2h;
Product distribution / selectivity;
|
98.3% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
copper(l) chloride;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 2h;
Product distribution / selectivity;
|
98.2% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
zinc(II) chloride;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 2h;
Product distribution / selectivity;
|
98.6% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
magnesium chloride;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 2h;
Product distribution / selectivity;
|
98.1% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)3ZnCl5;
In
1,2-dichloro-benzene;
at 20 - 175 ℃;
for 3h;
Product distribution / selectivity;
|
98.5% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
NH4MgCl3;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 2h;
Product distribution / selectivity;
|
98.2% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)2CoCl4;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 1.5h;
Product distribution / selectivity;
|
98.7% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)2CuCl4;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 2h;
Product distribution / selectivity;
|
98.4% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
xylene;
at 150 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
ammonium chloride; zinc(II) oxide;
In
xylene;
at 20 - 150 ℃;
for 7h;
Product distribution / selectivity;
|
98.1% |
|
phenol;
With
potassium hydroxide; sodium hydroxide;
In
xylene;
at 150 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)3ZnCl5;
In
xylene;
at 20 - 150 ℃;
for 8h;
Product distribution / selectivity;
Heating / reflux;
|
98.9% |
|
phenol;
With
potassium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
1,2-dichloro-benzene;
at 20 - 175 ℃;
for 2h;
Product distribution / selectivity;
|
98.5% |
|
phenol;
With
cesium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
ammonium chloride; zinc(II) oxide;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 1h;
Product distribution / selectivity;
|
98.1% |
|
phenol;
With
cesium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)3ZnCl5;
In
1,2-dichloro-benzene;
at 20 - 180 ℃;
for 1 - 3h;
Product distribution / selectivity;
Heating / reflux;
|
98.2% |
|
phenol;
With
cesium hydroxide; sodium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
1,2-dichloro-benzene;
at 20 - 175 ℃;
for 3h;
Product distribution / selectivity;
Heating / reflux;
|
98% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; potassium hydroxide;
In
chlorobenzene;
at 40 - 130 ℃;
for 6h;
Reagent/catalyst;
Solvent;
Temperature;
Inert atmosphere;
|
98% |
|
phenol;
With
calcium hydroxide; cesium hydroxide;
In
1,2-dichloro-benzene;
at 190 ℃;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
(NH4)3ZnCl5;
In
1,2-dichloro-benzene;
at 20 - 175 ℃;
for 3h;
Product distribution / selectivity;
|
97.6% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; sodium hydroxide;
for 0.0833333h;
Reagent/catalyst;
Microwave irradiation;
Green chemistry;
|
96.68% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; sodium hydroxide;
In
water; 1,1,2,2-tetrachloroethane;
at 145 ℃;
for 12h;
Temperature;
Solvent;
Autoclave;
|
96.81% |
|
phenol;
With
sodium hydroxide;
In
1,4-dioxane;
at 40 ℃;
for 1.5h;
Inert atmosphere;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
1,4-dioxane; diethylene glycol dimethyl ether;
at 115 ℃;
for 4.5h;
Solvent;
Temperature;
Reagent/catalyst;
Inert atmosphere;
|
93% |
|
With
pyridine; 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
chlorobenzene;
at 120 ℃;
for 12h;
|
92.5% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; tetrabutyl-ammonium chloride; sodium hydroxide;
In
water; chlorobenzene;
at 80 ℃;
Reagent/catalyst;
Solvent;
Temperature;
|
92% |
|
phenol;
With
sodium hydride;
In
tetrahydrofuran;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
tetrahydrofuran;
for 48h;
Reflux;
|
85% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; potassium carbonate;
In
chlorobenzene;
at 40 - 135 ℃;
Inert atmosphere;
|
82% |
|
With
potassium phosphate; 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
acetonitrile;
at 88 ℃;
for 10h;
Inert atmosphere;
|
80% |
|
With
potassium phosphate; 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
acetonitrile;
for 3h;
Heating;
|
75% |
|
With
sodium hydroxide; 2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
acetonitrile;
for 0.5h;
microwave irradiation;
|
70% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; caesium carbonate;
In
tetrahydrofuran;
at 20 ℃;
Inert atmosphere;
|
70% |
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; sodium hydride;
In
tetrahydrofuran; paraffin;
for 48h;
Yield given;
Heating;
|
|
|
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine; sodium hydride;
In
tetrahydrofuran;
Heating;
|
|
|
phenol;
With
sodium hydroxide;
In
toluene;
Azeotropic dehydration;
Heating / reflux;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
DMF (N,N-dimethyl-formamide); toluene;
at 80 ℃;
for 12h;
Product distribution / selectivity;
|
|
|
phenol;
With
sodium hydride;
In
tetrahydrofuran;
at 25 - 40 ℃;
for 0.5h;
Azeotropic dehydration;
Heating / reflux;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
tetrahydrofuran;
at 25 ℃;
for 24h;
Product distribution / selectivity;
|
|
|
phenol;
With
sodium hydroxide;
In
toluene;
Azeotropic dehydration;
Heating / reflux;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
DMF (N,N-dimethyl-formamide); toluene;
at 80 ℃;
for 12h;
Product distribution / selectivity;
|
|
|
phenol;
With
sodium hydroxide;
In
toluene;
Azeotropic dehydration;
Heating / reflux;
With
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine;
In
DMF (N,N-dimethyl-formamide); toluene;
at 80 ℃;
for 12h;
Product distribution / selectivity;
|
1184-10-7 Upstream products
-
139-02-6

sodium phenoxide
-
940-71-6
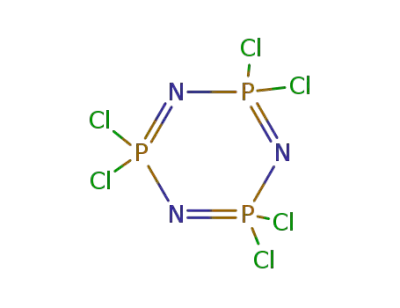
2,2,4,4,6,6-hexachloro-1,3,5-triaza-2,4,6-triphosphorine
-
2950-45-0
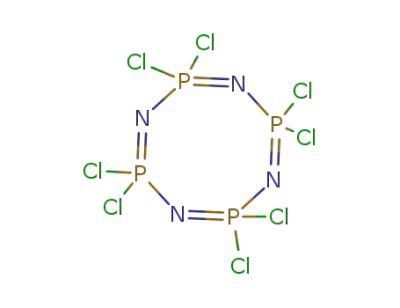
octachlorocyclotetraphosphazene
-
13596-41-3
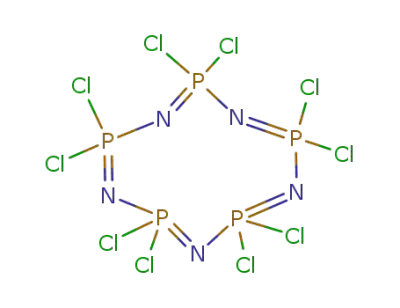
pentakisphosphorus nitride dichloride
1184-10-7 Downstream products
-
130551-84-7
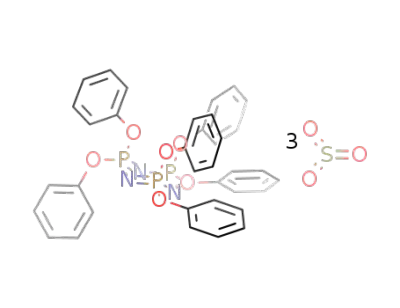
hexaphenoxycyclotriphosphazene * 3 sulfurtrioxide
-
134177-56-3
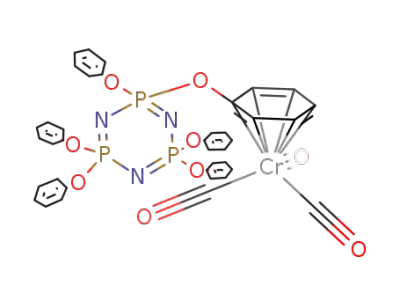
N3P3(OC6H5)5{(η6-OC6H5)Cr(CO)3}
-
130551-86-9
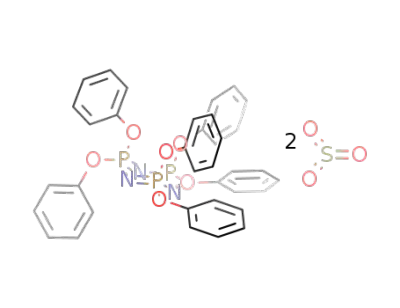
hexaphenoxycyclotriphosphazene * 2 sulfurtrioxide
-
1356917-82-2
N-methyl hexa(phenoxy)cyclotriphosphazenium trifluoromethanesulfonate
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
1-Bromo-4-chlorodibenzo[b,d]furan
CAS:2252237-87-7
-
4-bromo-1-chloro-Dibenzofuran
CAS:2183475-72-9