635-46-1
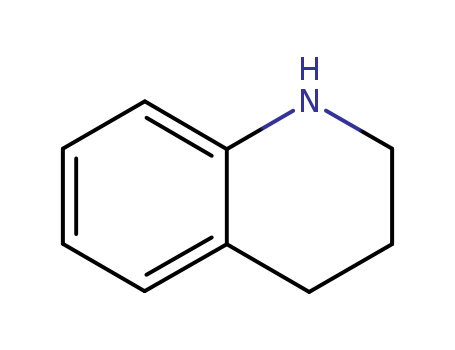
- Product Name:1,2,3,4-Tetrahydroquinoline
- Molecular Formula:C9H11N
- Purity:99%
- Molecular Weight:133.193
Product Details;
CasNo: 635-46-1
Molecular Formula: C9H11N
Appearance: clear pale yellow to yellow liquid
Quality Factory Supply 99% Pure 1,2,3,4-Tetrahydroquinoline 635-46-1 with Efficient Delivery
- Molecular Formula:C9H11N
- Molecular Weight:133.193
- Appearance/Colour:clear pale yellow to yellow liquid
- Vapor Pressure:0.0212mmHg at 25°C
- Melting Point:9-14 °C(lit.)
- Refractive Index:n20/D 1.593(lit.)
- Boiling Point:250.775 °C at 760 mmHg
- PKA:5.09±0.20(Predicted)
- Flash Point:100.6 °C
- PSA:12.03000
- Density:1.006 g/cm3
- LogP:2.18270
1,2,3,4-Tetrahydroquinoline(Cas 635-46-1) Usage
|
Synthesis Reference(s) |
Chemical and Pharmaceutical Bulletin, 34, p. 3905, 1986 DOI: 10.1248/cpb.34.3905Tetrahedron, 52, p. 1631, 1996 DOI: 10.1016/0040-4020(95)00991-4 |
|
General Description |
1,2,3,4-Tetrahydroquinoline is a bicyclic aromatic compound that serves as a substrate in enzymatic oxyfunctionalization reactions, yielding chiral synthons like (+)-(R)-1,2,3,4-tetrahydroquinoline-4-ol. It is also a key intermediate in hydrogenation processes, such as the selective reduction of quinoline using rhodium catalysts, and is utilized in the synthesis of heterocycles like epoxyisoindolo[2,1-a]quinolines and CETP inhibitors. Its derivatives exhibit potential pharmaceutical applications, including antihypoxic activity and modulation of lipid metabolism. |
|
Definition |
ChEBI: A member of the class of quinolines that is the 1,2,3,4-tetrahydro derivative of quinoline. |
InChI:InChI=1/C9H11N/c1-2-6-9-8(4-1)5-3-7-10-9/h1-2,4,6,10H,3,5,7H2
635-46-1 Relevant articles
Correction to: Halogen-Bonding-Induced Hydrogen Transfer to C=N Bond with Hantzsch Ester (Org. Lett. (2014) 16:12 (3244-3247) DOI: 10.1021/ol501259q)
He, Wei,Ge, Yi-Cen,Tan, Choon-Hong
, p. 7684 - 7684 (2019)
The structure of C1 has been revised in ...
Medium-Sized-Ring Analogues of Dibenzodiazepines by a Conformationally Induced Smiles Ring Expansion
Costil, Romain,Lefebvre, Quentin,Clayden, Jonathan
, p. 14602 - 14606 (2017)
Analogues of dibenzodiazepines, in which...
Study of Hydrodesulfurization by the Use of 35S-Labeled Dibenzothiophene. 2. Behavior of Sulfur in HDS, HDO, and HDN on Sulfided Mo/Al2O3 Catalyst
Kabe, Toshiaki,Quian, Weihua,Ishihara, Atsushi
, p. 912 - 916 (1994)
To investigate the behavior of sulfur du...
-
Tsodikov et al.
, (1967)
-
Ru-decorated N-doped carbon nanoflakes for selective hydrogenation of levulinic acid to γ-valerolactone and quinoline to tetrahydroquinoline with HCOOH in water
Chauhan, Arzoo,Kar, Ashish Kumar,Srivastava, Rajendra
, (2022/03/27)
The effective dissociation of biomass-de...
Dehydrogenative and Redox-Neutral N-Heterocyclization of Aminoalcohols Catalyzed by Manganese Pincer Complexes
Brzozowska, Aleksandra,Rueping, Magnus,Sklyaruk, Jan,Zubar, Viktoriia
, (2022/03/17)
A new manganese catalyzed heterocyclizat...
Chemoselective and Tandem Reduction of Arenes Using a Metal–Organic Framework-Supported Single-Site Cobalt Catalyst
Antil, Neha,Kumar, Ajay,Akhtar, Naved,Begum, Wahida,Chauhan, Manav,Newar, Rajashree,Rawat, Manhar Singh,Manna, Kuntal
supporting information, p. 1031 - 1040 (2022/01/19)
The development of heterogeneous, chemos...
Aromatic compound hydrogenation and hydrodeoxygenation method and application thereof
-
Paragraph 0094-0095; 0114-0122, (2021/05/29)
The invention belongs to the technical f...
635-46-1 Process route
-
-
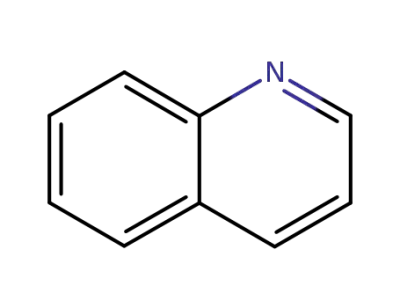
91-22-5
quinoline

-
-
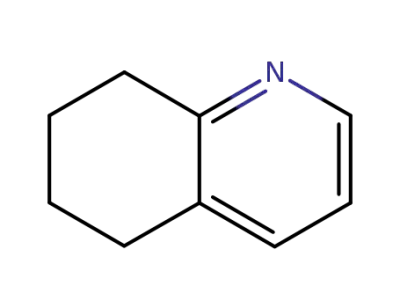
10500-57-9
5,6,7,8-tetrahydroquinoline

-
-
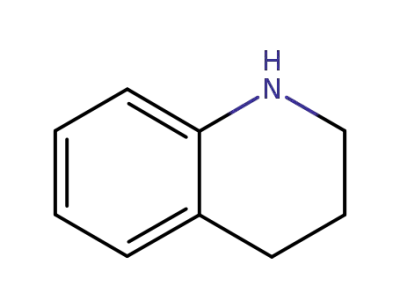
635-46-1
1,2,3,4-tetrahydroisoquinoline

-
-
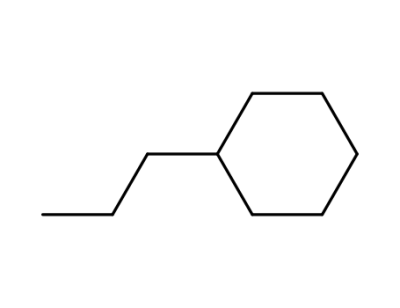
1678-92-8
propylcyclohexane

-
-
103-65-1
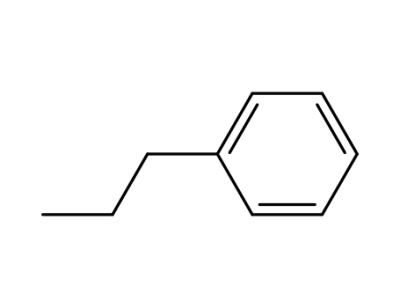
phenylpropane

-
-
1821-39-2
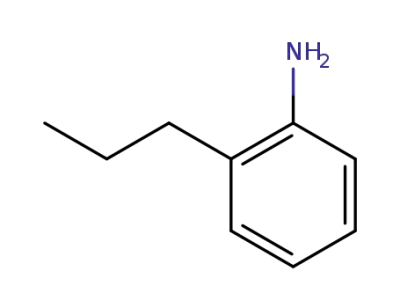
2-propylaniline
| Conditions | Yield |
|---|---|
|
With
hydrogen;
ruthenium sulphide on HY-zeolite;
In
n-heptane;
at 400 ℃;
for 4.5h;
under 25862.1 Torr;
Product distribution;
other catalysts;
|
-
-
50624-35-6
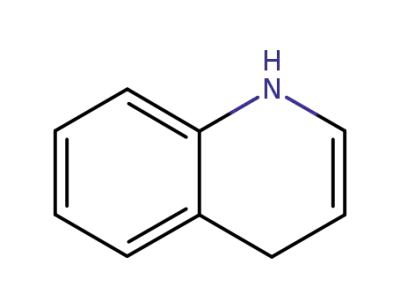
1,4-dihydroquinoline

-
-
1125-81-1
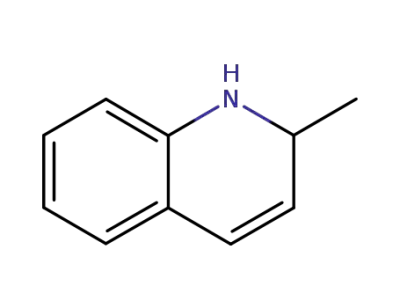
2-methyl-1,2-dihydroquinoline

-

-
635-46-1
1,2,3,4-tetrahydroisoquinoline

-
-
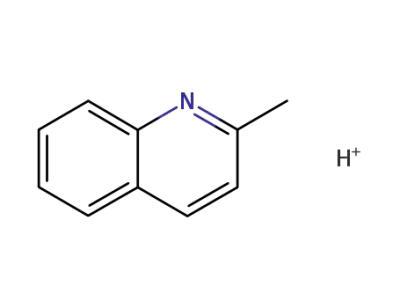
42307-21-1
2-Methyl-chinoliniumkation

-
-
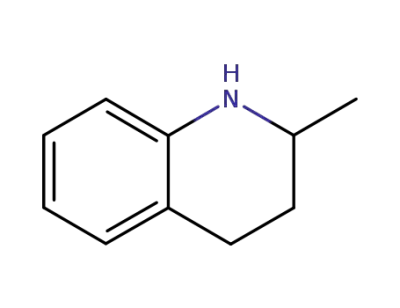
1780-19-4
1,2,3,4-tetrahydro-2-methylquinoline
| Conditions | Yield |
|---|---|
|
With
methanol;
hydrogenchloride;
Product distribution;
Mechanism;
Ambient temperature;
also with 6-methyl-1,2-dihydroquinoline substrate;
|
635-46-1 Upstream products
-
1466-88-2
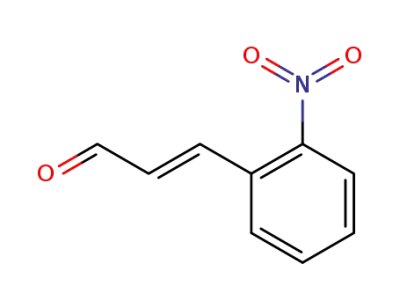
2-nitrocinnamic aldehyde
-
91-22-5

quinoline
-
2785-29-7
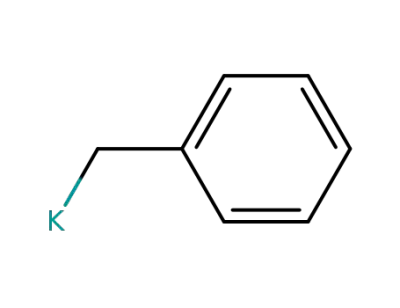
benzyl potassium
-
100-51-6
benzyl alcohol
635-46-1 Downstream products
-
21863-25-2
1-(1-methylethyl)-1,2,3,4-tetrahydroquinoline
-
3973-08-8
Thiazole-4-carboxylic acid
-
97460-75-8
(1,2,3,4-tetrahydro-quinolin-6-yl)-ethenetricarbonitrile
-
91-22-5

quinoline
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
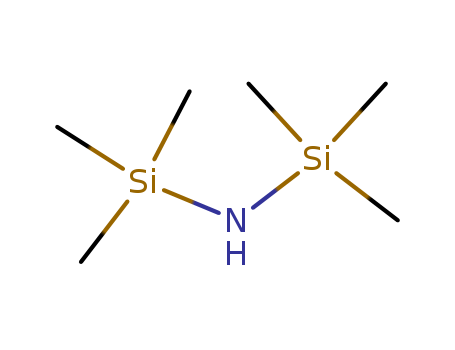
1,1,1,3,3,3-Hexamethyldisilazane
CAS:999-97-3
-
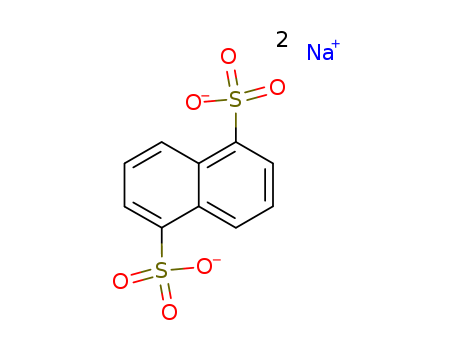
Disodium 1,5-naphthalenedisulfonate
CAS:1655-29-4