2842-44-6
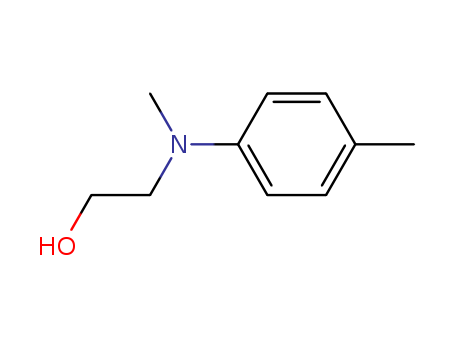
- Product Name:N-Methyl-N-hydroxyethyl-P-toluidine
- Molecular Formula:C10H15 N O
- Purity:99%
- Molecular Weight:165.235
Product Details;
CasNo: 2842-44-6
Molecular Formula: C10H15 N O
Factory Sells Best Quality N-Methyl-N-hydroxyethyl-P-toluidine 2842-44-6 with USP
- Molecular Formula:C10H15 N O
- Molecular Weight:165.235
- Vapor Pressure:0.00058mmHg at 25°C
- Boiling Point:298.1°Cat760mmHg
- PKA:14.69±0.10(Predicted)
- Flash Point:148.8°C
- PSA:23.47000
- Density:1.047g/cm3
- LogP:1.42350
N-(2-HYDROXYETHYL)-N-METHYL-4-TOLUIDINE(Cas 2842-44-6) Usage
|
Synthesis |
Add reactant (0.2 mmol, 1.0 equiv), THF (2.0 mL) and CH2Br2?(0.6 mmol, 3.0 equiv) to a flame-dried 10 mL Schlenk tube in a glove box. Seal and take out of the glove box. Cool the reaction mixture to -78°C. Add nBuLi (0.56 mmol, 2.8 equiv) dropwise under N2?atmosphere within 3minutes. Stir the reaction at -78°C for 30 minutes and add ZnCl2?(0.1 mL, 0.5equiv, 1.0 M in Et2O). Allow the mixture to warm to room temperature and stir for 1 hour. Cool the mixture to 0°C. Add a premixture of H2O2?(30% in H2O, 0.5 mL) and NaOH (2.0 M, 1.0 mL). Stir the mixture at room temperature for another 1 hour and dilute with water (20 mL). Extract with DCM (30 mL x 2) and dry over Na2SO4. Filter and concentrate under vacuum. Purify the crude product by silica gel flash column chromatography to obtain product.?1H NMR (CDCl3, 500 MHz) δ 7.08 (d, J = 8.4 Hz, 2H), 6.78 (d, J = 8.4 Hz,2H), 3.80 (t, J = 5.6 Hz, 2H), 3.43 (t, J = 5.4 Hz, 2H), 2.93 (s, 3H), 2.28 (s, 3H), 2.01 (brs, 1H).?13C NMR (CDCl3, 125 MHz) δ 148.3, 129.8, 127.1, 114.0, 60.1, 56.2, 39.1, 20.4. Fig The synthetic method of N-(2-hydroxyethyl)-N-methyl-4-toluidine |
InChI:InChI=1/C10H15NO/c1-9-3-5-10(6-4-9)11(2)7-8-12/h3-6,12H,7-8H2,1-2H3
2842-44-6 Relevant articles
Aza-Matteson Reactions via Controlled Mono-and Double-Methylene Insertions into Nitrogen-Boron Bonds
Xie, Qiqiang,Dong, Guangbin
supporting information, p. 14422 - 14427 (2021/09/29)
Boron-homologation reactions represent a...
Ru-Catalyzed Switchable N-Hydroxyethylation and N-Acetonylation with Crude Glycerol
Xin, Zhuo,Jia, Le,Huang, Yuxing,Du, Chen-Xia,Li, Yuehui
, p. 2007 - 2011 (2020/03/19)
Highly efficient Ru-catalyzed selective ...
Direct hydroxyethylation of amines by carbohydrates: Via ruthenium catalysis
Jia, Le,Makha, Mohamed,Du, Chen-Xia,Quan, Zheng-Jun,Wang, Xi-Cun,Li, Yuehui
supporting information, p. 3127 - 3132 (2019/06/18)
An efficient and halogen-free catalytic ...
A Direct C-H/Ar-H Coupling Approach to Oxindoles, Thio-oxindoles, 3,4-Dihydro-1 H-quinolin-2-ones, and 1,2,3,4-Tetrahydroquinolines
Hurst, Timothy E.,Gorman, Ryan M.,Drouhin, Pauline,Perry, Alexis,Taylor, Richard J. K.
supporting information, p. 14063 - 14073 (2016/02/18)
A copper(II)-catalysed approach to oxind...
2842-44-6 Process route
-
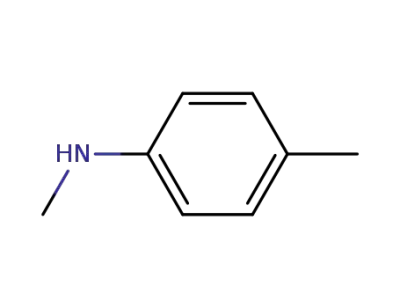
-
623-08-5
N-methyl-p-toluidine

-
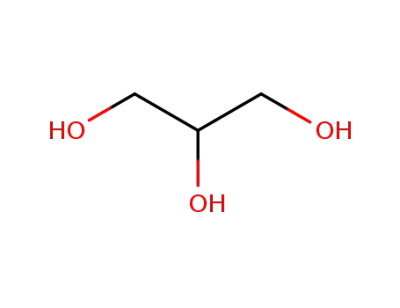
-
56-81-5,25618-55-7,64333-26-2,8013-25-0
glycerol

-
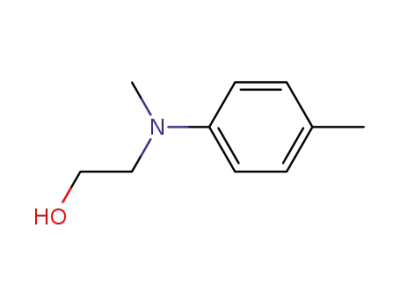
-
2842-44-6
2-(methyl(p-tolyl)amino)ethan-1-ol
| Conditions | Yield |
|---|---|
|
With
N-methyl-1-(thiophen-2-yl)methanamine; tris(triphenylphosphine)ruthenium(II) chloride; 1,1'-bis(dicyclohexylphosphinocyclopentadienyl)iron; potassium tert-butylate;
In
1,4-dioxane;
at 150 ℃;
for 20h;
|
91% |
-
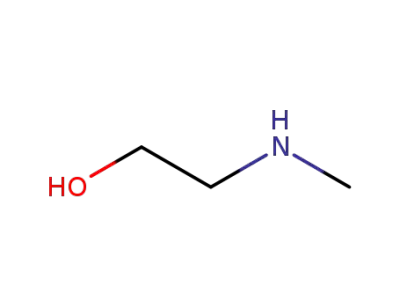
-
109-83-1
(2-hydroxyethyl)(methyl)amine

-
-
624-31-7
4-tolyl iodide

-

-
2842-44-6
2-(methyl(p-tolyl)amino)ethan-1-ol
| Conditions | Yield |
|---|---|
|
With
copper(l) iodide; sodium hydroxide;
In
water; dimethyl sulfoxide;
at 90 ℃;
for 16h;
Sealed tube;
Inert atmosphere;
|
82% |
2842-44-6 Upstream products
-
120547-24-2
N-(4-methylphenyl)-N-methylglycine ethyl ester
-
109-83-1

(2-hydroxyethyl)(methyl)amine
-
624-31-7
4-tolyl iodide
-
87-72-9
D-Xylose
2842-44-6 Downstream products
-
1262227-05-3
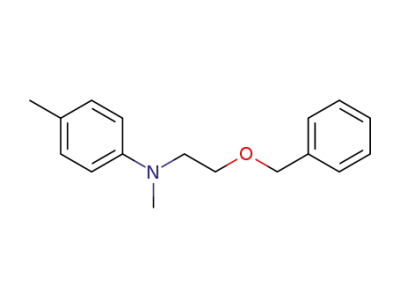
N-(2-(benzyloxy)ethyl)-N-methyl-p-toluidine
Relevant Products
-
benfooxythiamine
CAS:909542-99-0
-
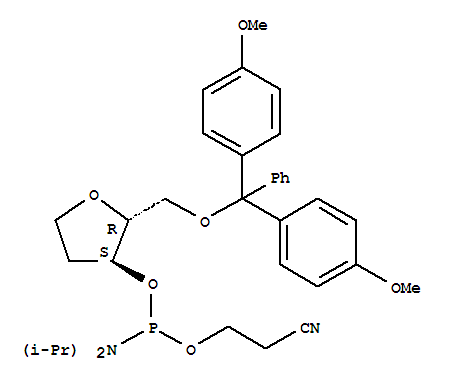
dSpacer CE Phosphoramidite
CAS:129821-76-7
-
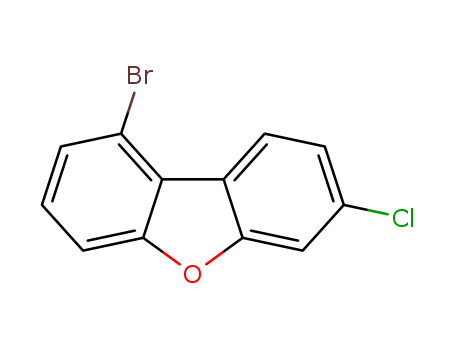
1-Bromo-7-chlorodibenzo[b,d]furan
CAS:2173555-52-5