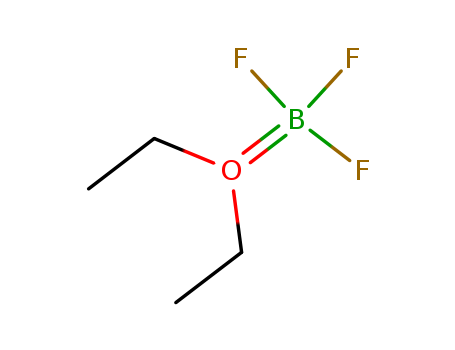
109-63-7
- Product Name:Boron trifluoride diethyl etherate
- Molecular Formula:C4H10BF3O
- Purity:99%
- Molecular Weight:141.929
Product Details;
CasNo: 109-63-7
Molecular Formula: C4H10BF3O
Appearance: colourless to brown fuming liquid with anacrid odour
Cost-effective and customizable Boron trifluoride diethyl etherate 109-63-7 in stock
- Molecular Formula:C4H10BF3O
- Molecular Weight:141.929
- Appearance/Colour:colourless to brown fuming liquid with anacrid odour
- Vapor Pressure:4.2 mm Hg ( 20 °C)
- Melting Point:-58 °C(lit.)
- Refractive Index:n20/D 1.344(lit.)
- Boiling Point:126 dec
- Flash Point:118°F
- PSA:17.07000
- Density:1.12 g/cm3
- LogP:1.92260
Boron trifluoride etherate(Cas 109-63-7) Usage
|
Chemical Description |
Boron trifluoride etherate is a Lewis acid catalyst used to obtain 1-(p_nitrophenyl)cyclopentane carboxaldehyde. |
|
uses |
Boron trifluoride diethyl etherate is used as a Lewis acid catalyst in Mukaiyama aldol addition, alkylation, acetylation, isomerization, dehydrations and condensation reactions. It is involved in the prepattion of polyethers in polymerization reactions. As a catalyst, it is used in the preparation of cyclopentyl- and cycloheptyl[b]indoles and other diborane. It is also used in sensitive neutron detectors in ionization chambers as well as monitoring radiation levels in earth?s atmosphere. |
|
Preparation |
Boron trifluoride gas, produced by heating the sulfuric acid, calcium fluoride (fluorite) and boric acid together, reacts with ether boron to produce the trifluoride etherate crude product, thus we can refine it to get the finished product. The consumption of raw material is as followed: boric acid (≥98%), 560kg/t; calcium fluoride(≥90%) 1150kg/t; fuming sulfuric acid (104.5%), 4100kg/t; ether(≥99%) 725kg/t.In absorption method shown in the chemical equations as followed, diethyl ether absorb boron trifluoride gas, produced by heating the sulfuric acid, calcium fluoride (fluorite) and boric acid together, to produce trifluoride etherate crude complex compound by vacuum distillation.3H2SO4+2H33BO3+3CaF2→2BF3+3CaSO4+6H2OBF3+(C2H5)2O→(C2H5)2O?BF3 |
|
Toxicity |
See boron trifluoride. |
|
Physical properties |
Fuming liquid; stable at ambient temperatures but hydrolyzed on exposure to moist air; density 1.125 g/mL; refractive index 1.348; solidifies at -60.4°C; boils at 125.7°C; flash point (open cup) 147°F (68.8°C); decomposes in water. |
|
Application |
Catalyst in the synthesis of polyol chains. Reagent for the coupling of imines to allylstannanes and 4′-nitrobenzenesulfenanilide to alkenes and alkynes.Lewis acid reagent with broad applicationCatalyst used in the preparation of cyclopentyl- and cycloheptyl[b]indoles from aryl cyclopropyl ketones via [3+2] cycloaddition. |
|
General Description |
Boron trifluoride etherate is a fuming liquid. Boron trifluoride etherate may be corrosive to skin, eyes and mucous membranes. Boron trifluoride etherate may be toxic by inhalation. Upon exposure to water Boron trifluoride etherate may emit flammable and corrosive vapors. Boron trifluoride etherate is used as a catalyst in chemical reactions. |
|
Air & Water Reactions |
Highly flammable. Fuming liquid, immediately hydrolyzed by moisture in air to form hydrogen fluoride [Merck 11th ed. 1989]. |
|
Reactivity Profile |
Boron trifluoride diethyl ether complex is a stable, highly flammable, colourless to brown fuming, corrosive liquid with a sharp pungent odour. It forms explosive peroxides in contact with air or oxygen. It reacts exothermically with water to form extremely flammable diethyl ether and toxic, corrosive boron trifluoride hydrates. The chemical is incompatible with bases, amines, and alkali metals. It immediately gets hydrolysed by moisture in air to form hydrogen fluoride. Boron trifluoride diethyl ether has applications in chemical laboratory as a catalyst in chemical reactions. |
|
Hazard |
The compound is highly toxic by inhalation. Skin contact causes burns. |
|
Health Hazard |
May cause toxic effects if inhaled or ingested/swallowed. Contact with substance may cause severe burns to skin and eyes. Fire will produce irritating, corrosive and/or toxic gases. Vapors may cause dizziness or suffocation. Runoff from fire control or dilution water may cause pollution. |
|
Fire Hazard |
Flammable/combustible material. May be ignited by heat, sparks or flames. Vapors may form explosive mixtures with air. Vapors may travel to source of ignition and flash back. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapor explosion hazard indoors, outdoors or in sewers. Runoff to sewer may create fire or explosion hazard. Containers may explode when heated. Many liquids are lighter than water. |
|
Flammability and Explosibility |
Flammable |
|
Potential Exposure |
Used as a catalyst. |
|
Shipping |
Diethyl: UN2604 Boron trifluoride diethyl etherate, Hazard class: 8; Labels: 8—Corrosive material, 3—Flammable liquid. Dimethyl: UN2965 Boron trifluoride dimethyl etherate, Hazard class: 4.3; Labels: 4.3— Dangerous when wet, 8—Corrosive material, 3— Flammable liquid. |
|
Incompatibilities |
Reacts with air forming corrosive hydrogen fluoride vapors. Incompatible with oxidizers (may cause fire and explosion), water, steam or heat, forming corrosive and flammable vapors. Peroxide containing etherate reacts explosively with aluminum lithium hydride, magnesium tetrahydroaluminate. Mixtures with phenol react explosively with 1,3-butadiene. Presumed to form explosive peroxides. |
InChI:InChI:1S/C4H10BF3O/c1-3-9(4-2)5(6,7)8/h3-4H2,1-2H3
109-63-7 Relevant articles
-
Seel
, p. 331,349 (1943)
-
Continuous synthesis method and reaction device of boron trifluoride complex
-
Paragraph 0067-0075, (2021/10/05)
The invention relates to the technical f...
Synthesis and reactivity studies of dicationic dihydrogen complexes bearing sulfur-donor ligands: A combined experimental and computational study
Gandhi, Thirumanavelan,Rajkumar, Subramani,Prathyusha,Priyakumar, U. Deva
, p. 1434 - 1443 (2013/05/22)
A series of dihydrogen complexes trans-[...
Synthesis and characterisation of some new boron compounds containing the 2,4,6-(CF3)3C6 H2 (fluoromes = Ar), 2,6-(CF3)2C6H3 (fluoroxyl = Ar′), or 2,4-(CF3)2 C6H3 (Ar″) ligands
Cornet, Stephanie M.,Dillon, Keith B.,Entwistle, Christopher D.,Fox, Mark A.,Goeta, Andres E.,Goodwin, Helen P.,Marder, Todd B.,Thompson, Amber L.
, p. 4395 - 4405 (2007/10/03)
Several new boron compounds containing t...
Syntheses, structures, and reactivity studies of half-open ruthenocenes and their oxodienyl analogues
Navarro Clemente, M. Elena,Saavedra, Patricia Juárez,Vásquez, Marisol Cervantes,Angeles Paz-Sandoval,Arif, Atta M.,Ernst, Richard D.
, p. 592 - 605 (2008/10/08)
Improved synthetic routes to Cp*Ru(Pdl) ...
109-63-7 Process route
-
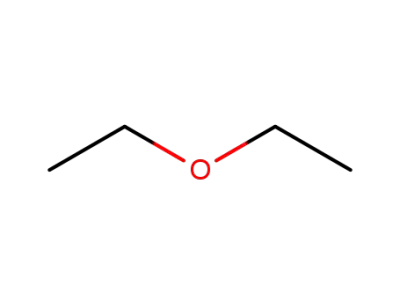
-
60-29-7,927820-24-4
diethyl ether

-
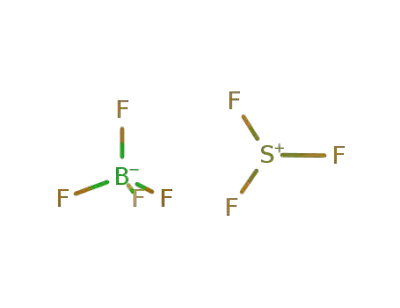
-
35963-96-3
Trifluorsulfonium-tetrafluoroborat

-

-
7783-60-0
sulfur tetrafluoride

-
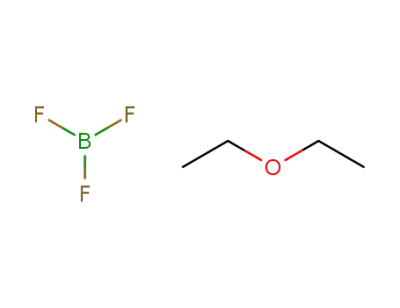
-
109-63-7
trifluoroborane diethyl ether
| Conditions | Yield |
|---|---|
|
|
|
|
|
-

-
60-29-7,927820-24-4
diethyl ether

-
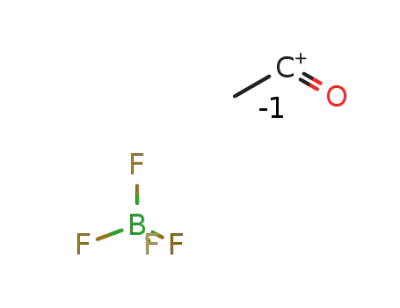
-
2261-02-1
acetylium tetrafluoroborate

-
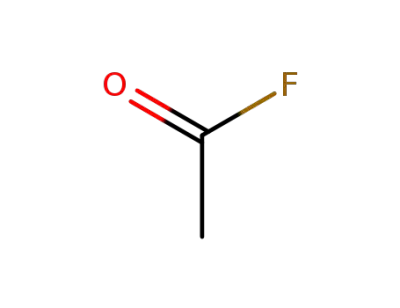
-
557-99-3
acetyl fluoride

-

-
109-63-7
trifluoroborane diethyl ether
| Conditions | Yield |
|---|---|
|
slight warming;
|
|
|
slight warming;
|
109-63-7 Upstream products
-
444-40-6
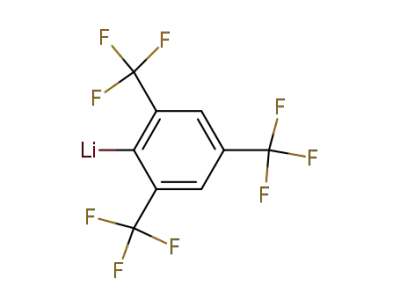
2,4,6-tris(trilfuoromethyl)phenyllithium
-
10294-34-5
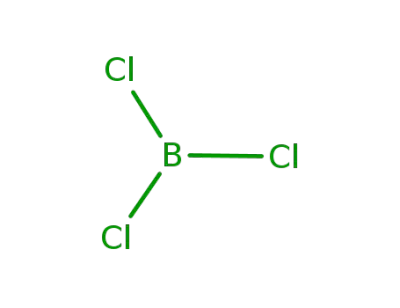
boron trichloride
-
60-29-7

diethyl ether
-
7637-07-2
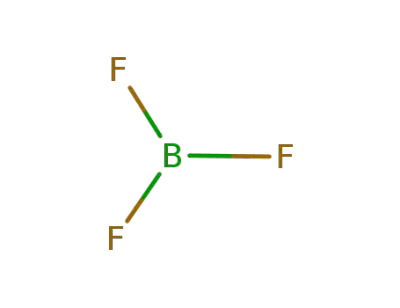
boron trifluoride
109-63-7 Downstream products
-
103185-78-0
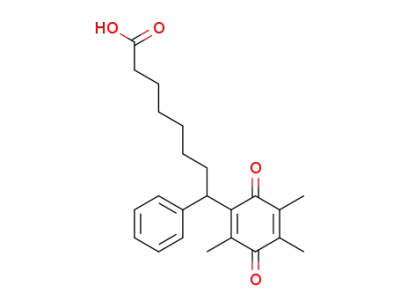
8-phenyl-8-(3,5,6-trimethyl-1,4-benzoquinon-2-yl)octanoic acid
-
110117-71-0
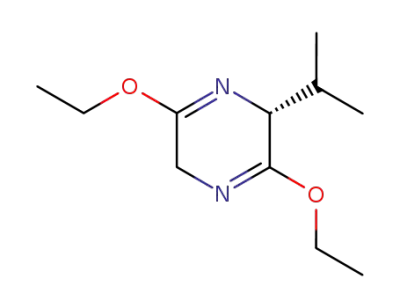
(2R)-3,6-diethoxy-2-isopropyl-2,5-dihydropyrazine
-
7298-21-7
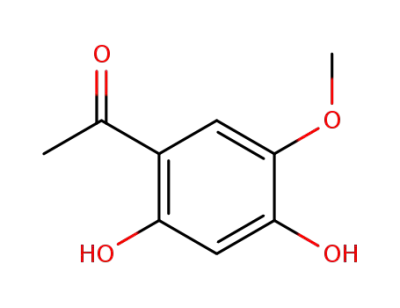
1-(2,4-dihydroxy-5-methoxyphenyl)ethan-1-one
-
90536-74-6
Ethyl 2,4,6-trihydroxybenzoate
Relevant Products
-
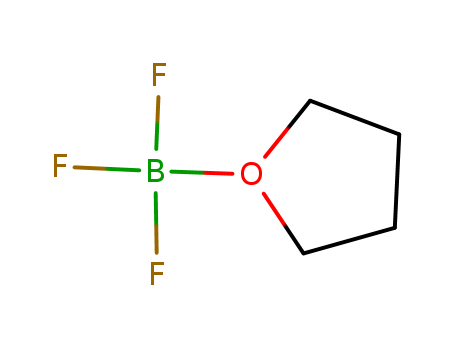
Boron Trifluoride Tetrahydrofuran
CAS:462-34-0
-
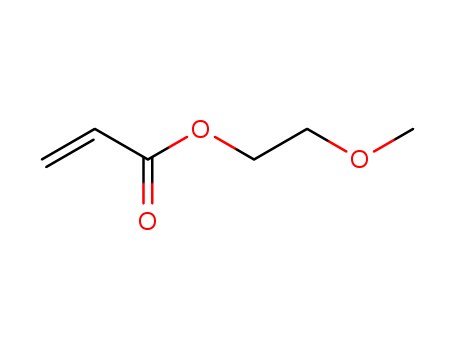
2-METHOXYETHYL ACRYLATE
CAS:3121-61-7
-
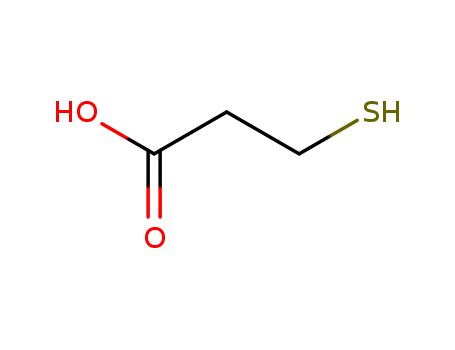
3-mercaptoproprionic acid
CAS:107-96-0